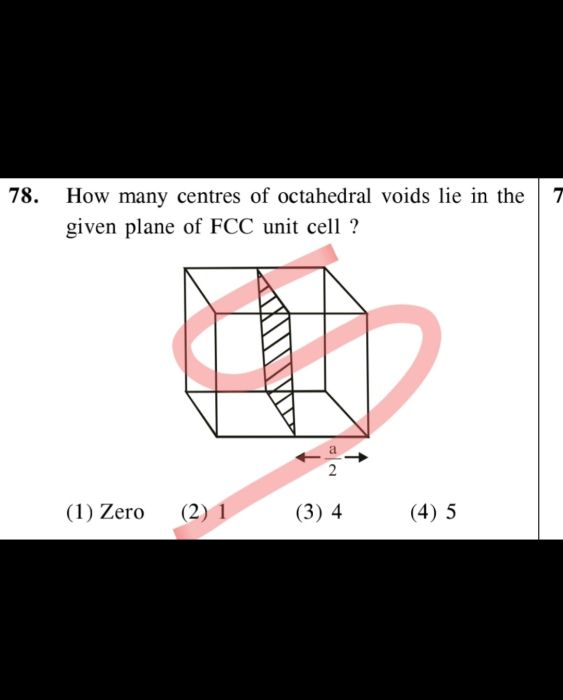
CBSE Class 12-science Answered
Why cubic unit cell cannot possess end-centred closed packing?? and what is the highest possible symmetry of this type?
Asked by vaish.agrawal | 26 Apr, 2015, 09:17: AM
In order to have end centered closed packing structure unit cell must possess vectors a≠b≠c, which is absent in case of cubic unit cell due to this cubic unit cell cannot possess end-centred closed packing. The highest symmetry possible for an end centered unit cell is tetragonal.
Answered by Arvind Diwale | 29 Apr, 2015, 10:51: AM
Concept Videos
CBSE 12-science - Chemistry
Asked by harshul2019 | 25 May, 2022, 08:52: PM
CBSE 12-science - Chemistry
Asked by arushidabhade | 17 Mar, 2021, 01:24: PM
CBSE 12-science - Chemistry
Asked by kasthurikalvi | 16 Sep, 2020, 03:46: PM
CBSE 12-science - Chemistry
Asked by sulaikhasulu393 | 01 Jul, 2020, 10:08: PM
CBSE 12-science - Chemistry
Asked by anukritisingh8103.bmps | 18 May, 2020, 03:00: PM
CBSE 12-science - Chemistry
Asked by rohitraman1115 | 10 Sep, 2019, 07:10: PM
CBSE 12-science - Chemistry
Asked by prakriti12oct | 05 Aug, 2019, 12:08: AM
CBSE 12-science - Chemistry
Asked by prakriti12oct | 04 Aug, 2019, 09:06: PM
CBSE 12-science - Chemistry
Asked by ranasingh04082002 | 31 Jul, 2019, 02:04: PM
CBSE 12-science - Chemistry
Asked by Saransekar407 | 11 Mar, 2019, 06:51: PM