CBSE Class 12-science Answered
why conc Hcl is used in reduction of nitro compounds instead oc conc sulphuric acid?
Asked by vasturushi | 26 Jun, 2017, 08:11: PM
Unlike hydrochloric acid, sulphuric acid is an oxidising agent. Hence, for reduction of nitro compounds, conc. HCl is used instead of conc. sulphuric acid.
Answered by Prachi Sawant | 28 Jun, 2017, 11:12: AM
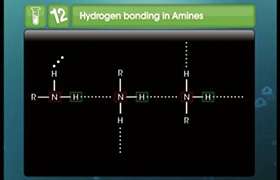
Concept Videos
CBSE 12-science - Chemistry
Asked by shwetayaligar205 | 07 Jul, 2022, 08:13: PM
CBSE 12-science - Chemistry
Asked by dhivagar25375 | 12 Aug, 2020, 08:34: PM
CBSE 12-science - Chemistry
Asked by danapalanandhan | 28 Jul, 2020, 11:48: AM
CBSE 12-science - Chemistry
Asked by ap996969 | 16 Mar, 2019, 10:17: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jul, 2014, 10:07: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jul, 2014, 10:30: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 06 Jun, 2016, 11:13: AM