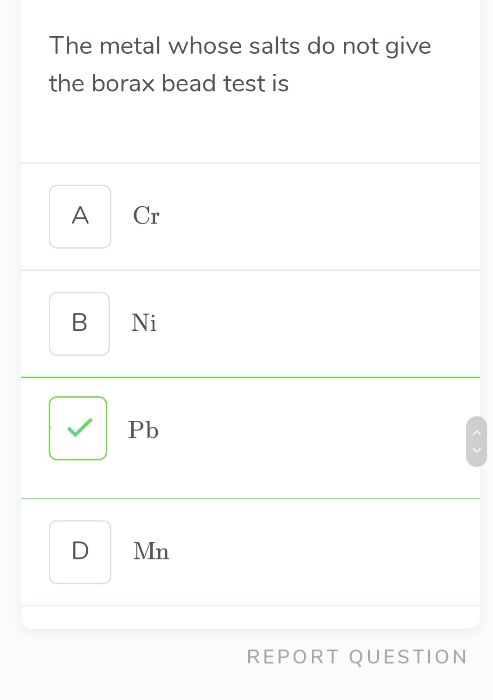
CBSE Class 11-science Answered
Why boron does not react with water?
Asked by Nitin Gupta | 22 Dec, 2012, 10:24: AM
Boron is a metalloid, exhibiting characteristics of both metals and non-metals. It is never found in its pure form in Earth, but it is a component of some minerals. However, pure boron can form as a result of chemical reactions. It can be a brown amorphous solid, or a crystalline solid. In the crystalline form, it is black and very hard. Its chemical properties are mostly, but not entirely, non-metallic. Like non-metallic compounds it doesnt react with water.
So, in pure mineral form and under normal conditions it doesn't react with water.
Answered by | 23 Dec, 2012, 08:44: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by singhnikita8717 | 05 Aug, 2021, 10:44: AM
CBSE 11-science - Chemistry
Asked by jaswindernkd | 21 Jun, 2020, 05:04: PM
CBSE 11-science - Chemistry
Asked by mufeedatvp2000 | 18 Apr, 2020, 02:21: PM
CBSE 11-science - Chemistry
Asked by Molaypaul700 | 10 Feb, 2020, 10:32: PM
CBSE 11-science - Chemistry
Asked by ABHILASHA | 08 Sep, 2019, 06:46: PM
CBSE 11-science - Chemistry
Asked by krishdabhoya2003 | 05 Aug, 2019, 08:19: AM
CBSE 11-science - Chemistry
Asked by vishakhachandan026 | 12 Jun, 2019, 09:20: AM
CBSE 11-science - Chemistry
Asked by pb_ckt | 28 Apr, 2019, 01:40: PM
CBSE 11-science - Chemistry
Asked by satya785583 | 16 Mar, 2019, 09:18: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 03 Jan, 2019, 01:09: PM