CBSE Class 12-science Answered
why are sulfonic acids stronger than carboxylic acids
Asked by neelsid | 09 May, 2011, 07:17: AM
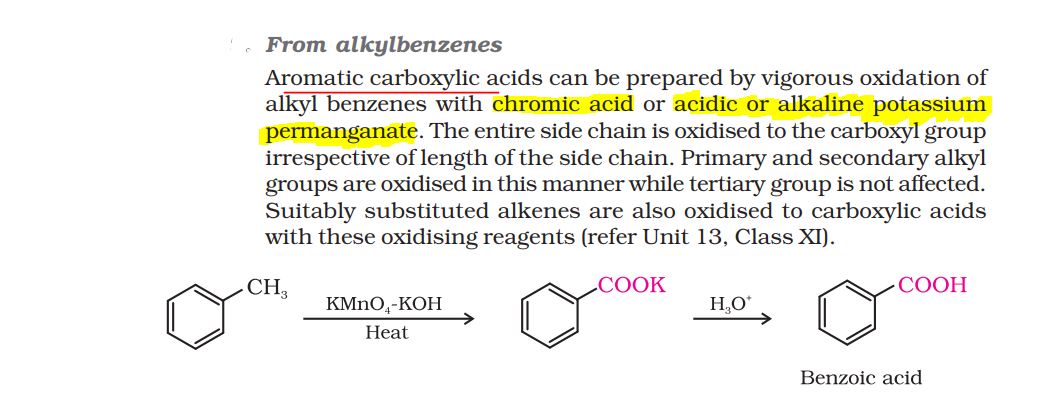
Sulphonic acids have general structure RSO2OH and carboxylic acids have structure RCOOH.
Strength of an acid depends on its tendency to undergo deprotonation to give the conjugate base of the acid .More stable is the conjugate base which in both of the above cases is a resonance stabilised anion, more will be the acidic stength.
In case of sulphonic acids the anion formed after deprotonation (RSO2O-) is more stable due to the resonsnce stabilisation of the anion by dispersal of the negative charge over three oxygen atoms present as compared to carboxylic acids in which the negative charge in the anion(RCOO-) is dispersed over only two oxygen atoms present.
Hence sulphonic acids are stonger acids than caboxylic acids.
Answered by | 10 May, 2011, 12:28: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by ukg8612 | 15 Apr, 2024, 07:36: PM
CBSE 12-science - Chemistry
Asked by ajayarchi | 08 Feb, 2024, 03:43: AM
CBSE 12-science - Chemistry
Asked by pallasriramulu9 | 24 Dec, 2023, 06:05: AM
CBSE 12-science - Chemistry
Asked by bsaheliya | 22 Dec, 2023, 09:53: PM
CBSE 12-science - Chemistry
Asked by ygarg8323 | 18 Apr, 2022, 12:47: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 30 Jun, 2021, 04:52: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 29 Jun, 2021, 08:36: AM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 28 Jun, 2021, 02:34: PM
CBSE 12-science - Chemistry
Asked by saimerala007 | 22 May, 2021, 02:08: PM
CBSE 12-science - Chemistry
Asked by amitkumar.cis | 31 Dec, 2020, 10:45: AM