CBSE Class 10 Answered
Which of the following is a redox reaction? (A) NaCl + KNO3 → NaNO3 + KCl
(B) CaC2O4 + 2HCl → CaCl2 + H2C2O4
(C) Mg(OH)2 + 2NH2Cl → MgCl2 + 2NH4OH (D) Zn + 2AgCN → 2Ag + Zn(CN)2
Asked by debjit_dm | 01 May, 2020, 04:41: PM
Following reaction is a redox reaction:
Zn + 2AgCN → 2Ag + Zn(CN)2
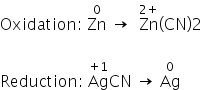
Answered by Ramandeep | 01 May, 2020, 06:44: PM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by ten.foundation | 29 Aug, 2022, 07:53: PM
CBSE 10 - Chemistry
Asked by bhuyanagas | 30 Nov, 2021, 09:46: PM
CBSE 10 - Chemistry
Asked by s158410a.niraj007031 | 03 Nov, 2021, 05:04: PM
CBSE 10 - Chemistry
Asked by shlok4559 | 01 Nov, 2021, 09:39: PM
CBSE 10 - Chemistry
Asked by pardeep360sehlang | 06 Oct, 2021, 09:28: PM
CBSE 10 - Chemistry
Asked by yogipharma11 | 22 Oct, 2020, 06:28: PM
CBSE 10 - Chemistry
Asked by nehchalrs15 | 02 Oct, 2020, 10:14: AM
CBSE 10 - Chemistry
Asked by nirupathak2004 | 21 May, 2020, 06:09: PM
CBSE 10 - Chemistry
Asked by bhuvaneswari5781 | 18 May, 2020, 11:13: AM
CBSE 10 - Chemistry
Asked by debjit_dm | 01 May, 2020, 04:41: PM









