JEE Class main Answered
What volume of a solution of Hcl containing 73g/litre would suffice for the exact nuetralisation of NaOH obtained by allowing 0.46g of metallic sodium to react with water?
Asked by goyatvanshika55 | 15 Jun, 2019, 06:36: PM
The reactions are
Na + H2O → NaOH + 1/2 H2
NaOH + HCl → NaCl + H2O
Meq of HCl = Meq of NaOH = Meq of Na
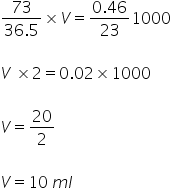
Volume required is 10 ml
Answered by Varsha | 16 Jun, 2019, 08:54: AM
JEE main - Chemistry
Asked by gmafia618 | 04 Apr, 2024, 08:48: PM
JEE main - Chemistry
Asked by jadhavshivtej256 | 27 Feb, 2024, 06:25: PM
JEE main - Chemistry
Asked by pradumankumarsah1 | 30 Jan, 2024, 02:36: PM
JEE main - Chemistry
Asked by srujan11042008 | 06 Nov, 2023, 10:31: AM
JEE main - Chemistry
Asked by vuppulojusaritha | 05 Nov, 2023, 02:22: PM
JEE main - Chemistry
Asked by radheshyambaheti085 | 09 Aug, 2023, 07:10: AM
JEE main - Chemistry
Asked by | 17 Aug, 2022, 08:10: PM
JEE main - Chemistry
Asked by aryankatiyar223 | 10 Aug, 2022, 11:57: PM
JEE main - Chemistry
Asked by prajwalravisharma | 01 May, 2022, 07:12: AM



