ICSE Class 9 Answered
What temperature would be necessary to double the volume of a gas initially at s.t.p. if the pressure is decreased by 50% ?
Ans [0℃]
Asked by aadrica.kanika | 27 Dec, 2018, 12:27: PM
Given:
At STP
P1 =760 mmHg
V1 = V1
T1 = 273 K
P2 = 50% OF P1
= 380 mmHg
V2 =2V1
T1 = ?
We have,
Gas equation,
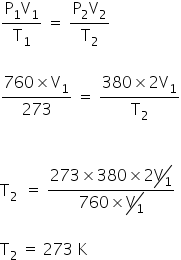
The temperature necessary to double the volume of a gas is 273 K or 0 °C.
Answered by Varsha | 28 Dec, 2018, 11:07: AM
Concept Videos
ICSE 9 - Chemistry
Asked by zainaali39692 | 04 Dec, 2020, 08:53: AM
ICSE 9 - Chemistry
Asked by gup.navya2006 | 01 Dec, 2020, 09:28: AM
ICSE 9 - Chemistry
Asked by Vishusingh2020.2021 | 25 Sep, 2020, 10:09: PM
ICSE 9 - Chemistry
Asked by sudesghnapattanayak2017 | 19 May, 2020, 08:13: PM
ICSE 9 - Chemistry
Asked by abeshchakraborty6 | 23 Feb, 2020, 08:54: AM
ICSE 9 - Chemistry
Asked by dnlwalkers | 08 Jan, 2020, 09:57: AM
ICSE 9 - Chemistry
Asked by raichuratanvi | 14 Dec, 2019, 12:02: PM
ICSE 9 - Chemistry
Asked by merajanjum87 | 21 Nov, 2019, 09:53: PM
ICSE 9 - Chemistry
Asked by parvathimanjunath24 | 31 Oct, 2019, 09:34: PM
ICSE 9 - Chemistry
Asked by devbhaisha.tl | 21 Sep, 2019, 10:02: PM



