CBSE Class 11-science Answered
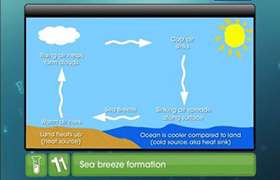
The second law of thermodynamics introduces the concept of entropy and its relation with spontaneous processes. In an isolated system such as mixing of gases, there is no exchange of energy or matter between the system and the surroundings. But due to increase in randomness, there is increase in entropy. Thus, it can be stated that for a spontaneous process in an isolated system, the change in entropy is positive or DS > 0. However, if the system is not isolated, the entropy changes of the system and the surroundings are also to be taken into account. Then, the total entropy change (DStotal) will be equal to the sum of the change in entropy of the system (DSsystem) and the change in entropy of the surroundings (DSsurroundings).
DStotal = DSsystem + DSsurroundings
For a spontaneous process, DStotal must be positive;
DStotal = DSsystem + DSsurroundings > 0
But system and surroundings constitute universe for thermodynamic point of view so that for spontaneous change, DSuniverse > 0.