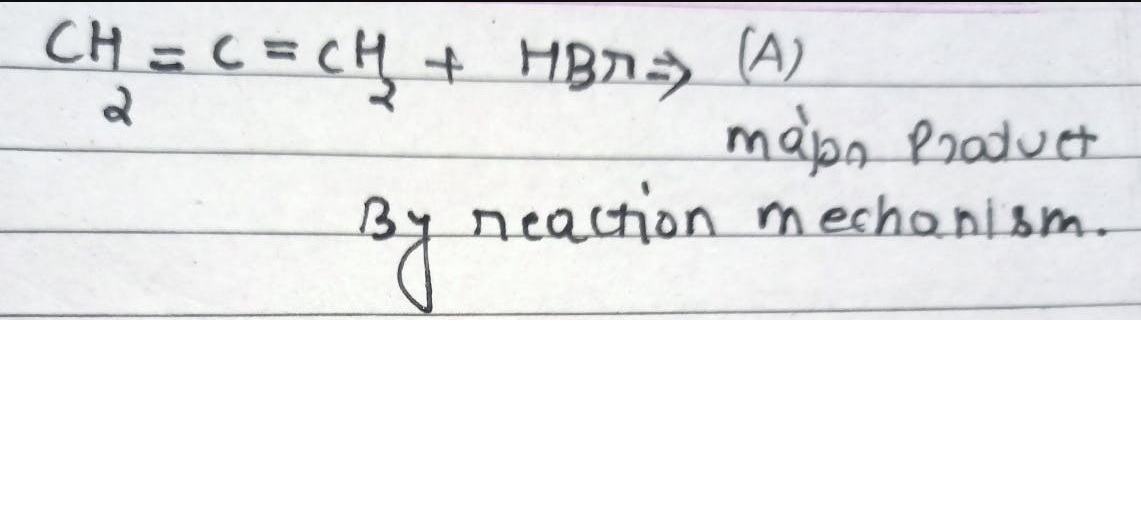CBSE Class 12-science Answered
what is the main product??
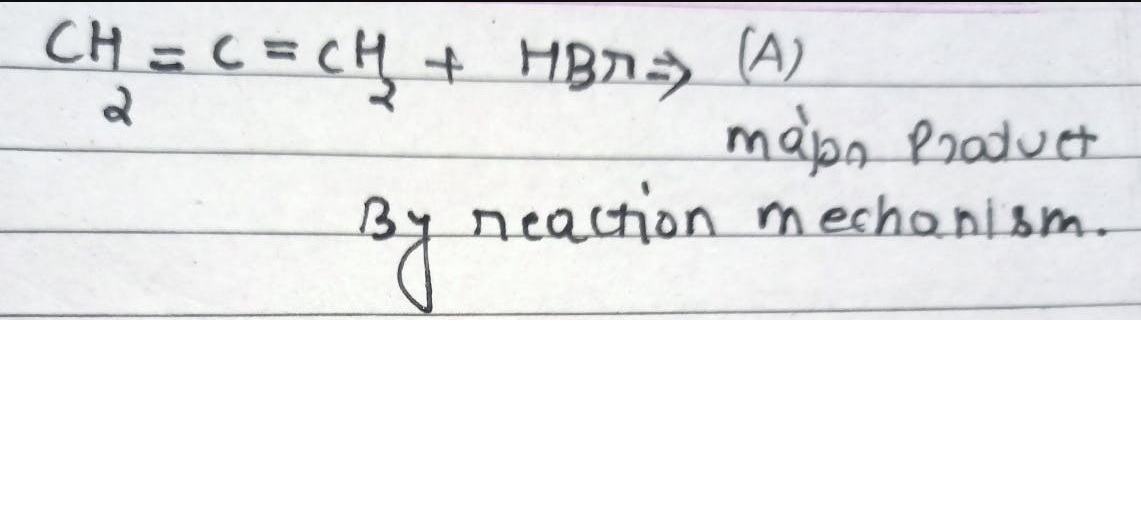
Asked by amitkumar.cis | 01 Jan, 2021, 09:15: PM
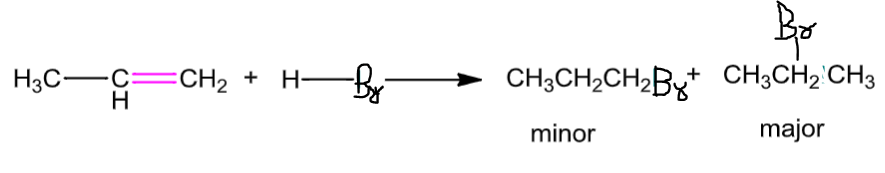
Addition of hydrogen halides: An alkene is converted to corresponding alkyl halide by reaction with hydrogen chloride, hydrogen bromide or hydrogen iodide
Propene yields two products; however,only one predominates according toMarkovnikov’s rule.
Propene yields two products; however,only one predominates according toMarkovnikov’s rule.
The rule states that,
“The addition of unsymmetrical reagents such as HX, H2O, HOX, etc. to unsymmetrical alkenes occurs in such a way that the negative part of the addendum (i.e., adding molecule) goes to that carbon atom of the double bond which carries lesser number of hydrogen atoms.”
Answered by Ramandeep | 01 Jan, 2021, 10:27: PM
Application Videos
Concept Videos
CBSE 12-science - Chemistry
Asked by surajbhanupatro44 | 07 Nov, 2023, 12:01: AM
CBSE 12-science - Chemistry
Asked by mayamishra9540500880 | 04 Jul, 2022, 07:11: PM
CBSE 12-science - Chemistry
Asked by harshaldpathak | 11 Jun, 2022, 05:37: PM
CBSE 12-science - Chemistry
Asked by amitkumar.cis | 01 Jan, 2021, 09:15: PM
CBSE 12-science - Chemistry
Asked by me.mirzainayat | 14 Nov, 2020, 07:31: AM
CBSE 12-science - Chemistry
Asked by Prachidewangan74 | 02 Oct, 2020, 03:02: PM
CBSE 12-science - Chemistry
Asked by sujithanathan119 | 01 Jun, 2020, 12:00: PM
CBSE 12-science - Chemistry
Asked by ng9045007209 | 21 May, 2020, 07:47: PM
CBSE 12-science - Chemistry
Asked by gangavaramouni | 26 Mar, 2020, 10:33: AM