CBSE Class 12-science Answered
what is the difference between frequency and intensity of light
Asked by lakshmiprasadsingh | 18 Feb, 2014, 07:09: AM
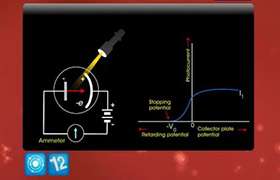
The energy of a photon is determined by its frequency, and the intensity of light is determined by the number of photons.
In photoelectric effect, when a photon strikes a metal surface photo electrons are emitted from the surface. Electrons are ejected only if its energy is greater than the work function of the metal. Hence, if the frequency of the light is too low, then it doesn't matter how intense the light is, since the energy of the photon is lower than the work function.
What the intensity does change is the number of photoelectrons emitted. The more photons (Intensity), the more electrons struck and the more that are ejected.
Hence, the energy of emitted electrons is directly proportional to the frequency of the radiation and independent of intensity of the radiation.
Answered by | 18 Feb, 2014, 11:08: AM
Concept Videos
CBSE 12-science - Physics
Asked by mishrigupta19319 | 08 Apr, 2024, 06:28: PM
CBSE 12-science - Physics
Asked by mishrigupta19319 | 07 Apr, 2024, 11:23: AM
CBSE 12-science - Physics
Asked by mazhartahsildar143 | 07 May, 2020, 01:44: PM
CBSE 12-science - Physics
Asked by Hemanthrakshitha95 | 03 Mar, 2020, 02:55: PM
CBSE 12-science - Physics
Asked by shivakumarshreyas24 | 01 Mar, 2020, 08:12: AM
CBSE 12-science - Physics
Asked by khushimassey437 | 31 May, 2019, 08:41: AM
CBSE 12-science - Physics
Asked by jain.pradeep | 26 May, 2019, 06:42: PM
CBSE 12-science - Physics
Asked by manasvijha | 19 Mar, 2019, 07:17: PM
CBSE 12-science - Physics
Asked by rohitraman1115 | 08 Jan, 2019, 04:33: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 21 May, 2014, 09:18: AM