CBSE Class 11-science Answered
What is the difference between adding inert gas at constant volume and adding inert gas at constant pressure?
Asked by Benjamin | 03 Sep, 2015, 06:01: PM
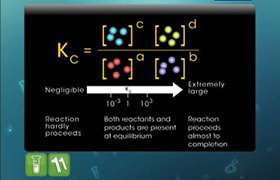
At equilibrium,
When an inert gas is added at constant volume, the pressure increases. But the ratio of the moles of reactants and products to the volume does not change.
Hence, there will nit be any effect on the equilibrium of the reaction.
When the inert gas is added at constant pressure, the total volume will increase. Thus, the number of moles of the reactants and products per unit volume will drcrease. That means the concentration of the reactants and products will be changed. Hence, the equilibrium will shift towards the direction where there is an increase in the number of moles of gases.
Example:
2NH3(g) ↔ N2(g) + 3H2(g)
In the above case, addition of inert gas at constant pressure, will shift the equilibrium towards product side as the total number of moles of the products is more.
Answered by Prachi Sawant | 04 Sep, 2015, 10:43: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by visank90 | 24 Nov, 2023, 10:45: AM
CBSE 11-science - Chemistry
Asked by rakhikumarithakur4 | 22 May, 2020, 08:21: PM
CBSE 11-science - Chemistry
Asked by arshrana3272 | 04 Mar, 2020, 02:57: PM
CBSE 11-science - Chemistry
Asked by hsdhall.2005 | 12 Nov, 2019, 11:40: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 19 Jul, 2018, 08:52: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 16 Jun, 2016, 05:17: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 16 Jun, 2016, 05:17: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 27 Apr, 2015, 03:08: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 27 Apr, 2015, 04:12: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 27 Apr, 2015, 04:15: PM




 6H2O (g) + 2N2 (g) How will the equilibrium shift if: a) The volume is increased, b) Helium gas is added
6H2O (g) + 2N2 (g) How will the equilibrium shift if: a) The volume is increased, b) Helium gas is added