CBSE Class 12-science Answered
What is Saponification process?
Asked by Topperlearning User | 09 Jul, 2014, 02:14: PM
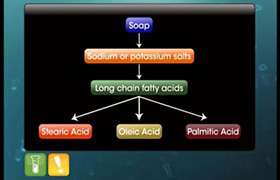
Soaps containing sodium salts are formed by heating fat i.e. glyceryl ester of fatty acids with aqueous sodium hydroxide solution. The reaction is known as Saponification reaction. In such reaction esters of fatty acids are hydolysed and the soap remains in colloidal form. It is precipitated from the solution by adding sodium chloride. The solution left after removing the soap contains Glycerol.
Answered by | 09 Jul, 2014, 04:14: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by Topperlearning User | 09 Jul, 2014, 02:14: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 22 Jun, 2016, 10:50: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 09 Jul, 2014, 02:17: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 22 Jun, 2016, 10:52: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 09 Jul, 2014, 02:20: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 09 Jul, 2014, 04:54: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 09 Jul, 2014, 04:56: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 09 Jul, 2014, 05:03: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 22 Jun, 2016, 10:52: AM