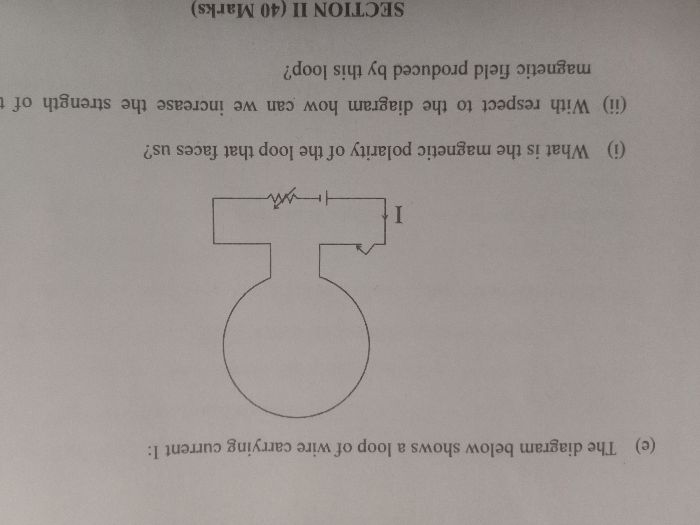
ICSE Class 10 Answered
Polarity is defined as the property in a molecule, or compound through which they are either attracted or repelled by an electric charge because of an asymmetrical arrangement of electropositive or electronegative atoms around the center of the species.
In simple words, polarity is the unsymmetrical distribution of charge on a molecule or compound so that there arises a negative and a positive pole. The species having polarity are called as dipole and must have a resultant dipole moment.
The best example of polarity is found in ionic compounds which have a negative and a positive charge centers and in some covalent compounds too which have difference in the electro-negativity of attached groups. These covalent compounds are known as polar covalent compounds.