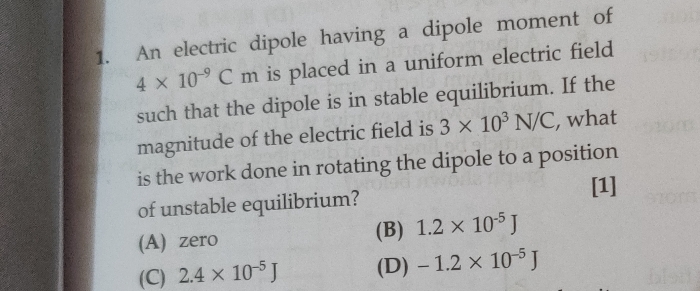CBSE Class 12-science Answered
What is an ideal electric dipole and what is meant by a permanent dipole?
Asked by NAKSHATRA | 25 Jul, 2014, 08:18: AM
A pair of equal and opposite charges seperated by a small vector distance is called an electric dipole.
An ideal dipole consists of two very very large charges + q and - q seperated by a very very small distance.
An ideal dipole has almost no size.
Molecules are made up of positive and negative charges seperated by small distances like water, ammonia etc and they act as electric dipoles.
Molecules which have non uniform distribution of charge are known as polar.Polar molecules will always be slightly negative on one end and slightly positive on the other Polar molecules are said to be permanent dipoles.
Eg: water, carbon dioxide.
Answered by Jyothi Nair | 25 Jul, 2014, 09:19: AM
Concept Videos
CBSE 12-science - Physics
Asked by sachin.sondur2012 | 07 Feb, 2024, 11:26: AM
CBSE 12-science - Physics
Asked by millionairekishan | 25 Jan, 2024, 12:34: PM
CBSE 12-science - Physics
Asked by mahantabibhutibhusan261 | 06 Nov, 2023, 09:04: PM
CBSE 12-science - Physics
Asked by shinghip6 | 17 Jul, 2022, 04:36: PM
CBSE 12-science - Physics
Asked by bolojubakesri47 | 15 Jul, 2022, 06:15: PM
CBSE 12-science - Physics
Asked by surajsinghrajput4691 | 16 May, 2022, 12:20: AM
CBSE 12-science - Physics
Asked by CHITTIBOMMAPARDHANADH | 06 Sep, 2021, 02:27: PM
CBSE 12-science - Physics
Asked by krishnamm7000 | 25 Jun, 2021, 07:30: AM
CBSE 12-science - Physics
Asked by KRISHPATEL.soc | 20 Jun, 2021, 02:02: PM