CBSE Class 11-science Answered
What are the enthalpies of the elements and what are the standard states of enthalpies elements?
* Unity
* Zero
* Negative
* Positive
Asked by shibu mann | 07 Dec, 2010, 12:00: AM
The enthalpy of formation depends upon the conditions of formation. i.e. temperature, pressure and physical state (gas solid or liquid) or allotropic state of the reactants. If all the reactants of a chemical reactions are in their standard state (i.e. at 25 degree C and 1 atmospheric pressure), the heat of formation or enthalpy of formation is called standard heat of formation or standard heat of formation.
By definition, the standard enthalpy (heat) of formation of an element in its standard state is zero,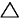 Hfo = 0.
Hfo = 0.
By definition, the standard enthalpy (heat) of formation of an element in its standard state is zero,
Answered by | 08 Dec, 2010, 08:10: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by rukayabatool395 | 24 Mar, 2024, 07:48: PM
CBSE 11-science - Chemistry
Asked by mankdubey670 | 06 Jun, 2022, 01:27: PM
CBSE 11-science - Chemistry
Asked by rama26516 | 12 Mar, 2022, 01:49: PM
CBSE 11-science - Chemistry
Asked by advssdrall | 11 Jan, 2022, 07:44: PM
CBSE 11-science - Chemistry
Asked by mossewalasindhu | 20 Nov, 2021, 02:16: PM
CBSE 11-science - Chemistry
Asked by chandankumargochhayat6 | 19 May, 2021, 08:48: AM
CBSE 11-science - Chemistry
Asked by paulnaveed202 | 17 Dec, 2020, 12:36: PM
CBSE 11-science - Chemistry
Asked by kjay0981 | 13 Dec, 2020, 03:45: PM
CBSE 11-science - Chemistry
Asked by adityasolanki7773 | 22 Oct, 2020, 03:40: PM
CBSE 11-science - Chemistry
Asked by pranavisrihari | 08 Sep, 2020, 05:24: PM