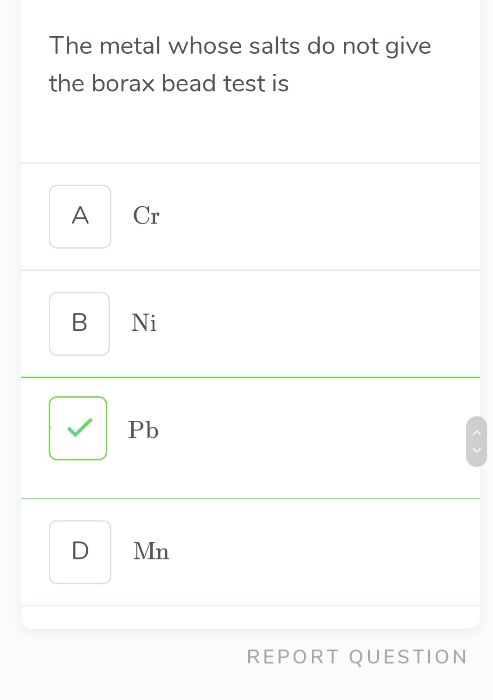
CBSE Class 11-science Answered
What are bcb and bb bonds? plz explain in detail......
Asked by Saurav Singh | 27 Jan, 2014, 11:15: PM
Structure of B2H6:
- The compound contains two hydrogen bridges in which four electrons are present, one electron from each boron atom and one electron from each hydrogen atom.
- Each of these bridges contains a B-H-B bond of two electrons, one from on B-atom and one from H-atom.
- These bonds are also called banana bond.
- Each boron atom which is sp2 hybridised is also attached with two H-atoms by covalent bond in such a way so that both B-atoms which are not attached with each other and all the four covalently bonded H-atoms lie in one plane.
- Out of two bridges bonds, each bond is placed above and below in a plane perpendicular to the plane of boron and terminal hydrogen atoms.
Answered by | 07 Apr, 2014, 12:13: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by singhnikita8717 | 05 Aug, 2021, 10:44: AM
CBSE 11-science - Chemistry
Asked by jaswindernkd | 21 Jun, 2020, 05:04: PM
CBSE 11-science - Chemistry
Asked by mufeedatvp2000 | 18 Apr, 2020, 02:21: PM
CBSE 11-science - Chemistry
Asked by Molaypaul700 | 10 Feb, 2020, 10:32: PM
CBSE 11-science - Chemistry
Asked by ABHILASHA | 08 Sep, 2019, 06:46: PM
CBSE 11-science - Chemistry
Asked by krishdabhoya2003 | 05 Aug, 2019, 08:19: AM
CBSE 11-science - Chemistry
Asked by vishakhachandan026 | 12 Jun, 2019, 09:20: AM
CBSE 11-science - Chemistry
Asked by pb_ckt | 28 Apr, 2019, 01:40: PM
CBSE 11-science - Chemistry
Asked by satya785583 | 16 Mar, 2019, 09:18: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 03 Jan, 2019, 01:09: PM