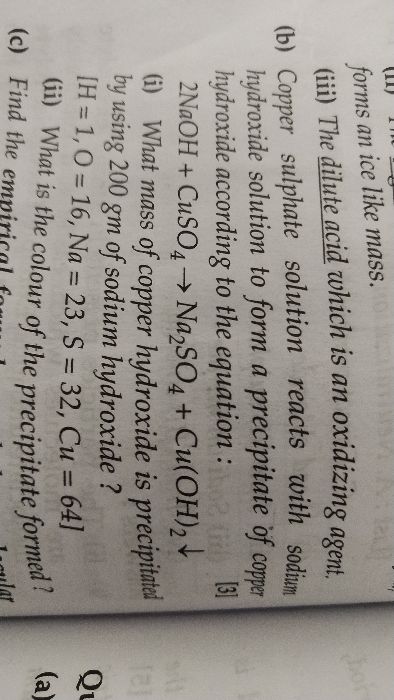ICSE Class 10 Answered
|
Mole and Gram Atomic Mass: One mole of atoms = 6.022 ×1023 atoms = Gram atomic mass of an element = 1 gram atom of the element
|
|
|
Mole and Gram Molecular Mass: One mole of molecules = 6.022 ×1023 molecules = Gram molecular mass = 1 gram molecule of the compound
|
|
|
Mole in terms of volume: One mole of a gas = 22.4 litres at S.T.P.
|
|
|
Moles of an element = Mass of the element Atomic mass or GA.W
|
Moles of a compound = Mass of the compound Molecular mass or GMW
|
|
Mass of one atom = Atomic Mass or GAW 6.022 × 1023
|
Mass of one molecule = Molecular Mass or GAW 6.022 × 1023
|
|
Number of molecules = Moles × 6.022 ×1023
|
Number of atoms = Moles × 6.022 ×1023
|