CBSE Class 12-science Answered
The rate of a reaction becomes four times when the temperature changes from 293 K to 313 K. Calculate the energy change of the reaction assuming that it does not change with temperature.
Asked by Topperlearning User | 22 Jun, 2016, 09:13: AM
T1 = 293 K; T2 = 313 K
According to Arrhenius equation:
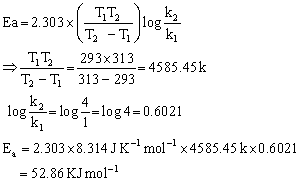
Answered by | 22 Jun, 2016, 11:13: AM
Concept Videos
CBSE 12-science - Chemistry
Asked by Topperlearning User | 22 Jun, 2016, 09:13: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 28 Mar, 2014, 01:38: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 22 Jun, 2016, 09:15: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 28 Mar, 2014, 01:39: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 22 Jun, 2016, 09:13: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 22 Jun, 2016, 09:13: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 28 Mar, 2014, 01:50: PM