CBSE Class 11-science Answered
State the factors on which mean free path (λ) of a gas molecule depends.
Asked by Topperlearning User | 11 May, 2015, 01:59: PM
The factors on which the mean free path (λ) of a gas molecule depends are:
(i) Diameter of the molecule: 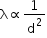
(ii) Number of gas molecules per unit volume: 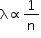
(iii) It depends indirectly on factors like temperature, pressure and Boltzmann constant.
Answered by | 11 May, 2015, 03:59: PM
Concept Videos
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 11 May, 2015, 01:59: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 11 May, 2015, 02:13: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 11 May, 2015, 02:26: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 11 May, 2015, 02:30: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM