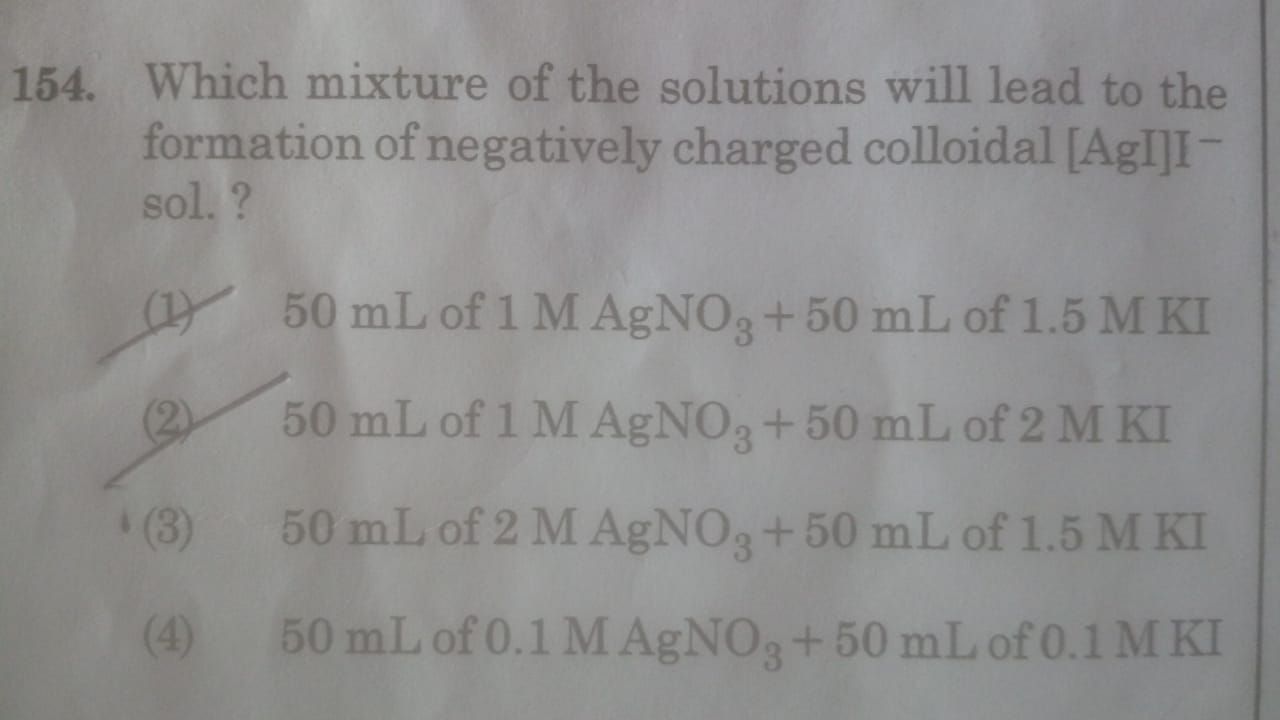CBSE Class 12-science Answered
sir plz explain the second sentence of the statement within semicolon("")
Adsorption is a surface phenomenon: During the phenomenon of
adsorption the adsorbate molecules are held on the surface of the
adsorbent by weak or strong (co- valent bond) forces.
"Therefore heat is
liberated during adsorption.
It is further more clear from the fact that finely divided adsorbents are good
adsorbents, because free forces increase on subdivision."
Asked by mohitgehlot | 03 Mar, 2011, 03:28: PM
When we powdered the adsorbent the active sites becomes more and more adsorbate can attached with the bonds.more bonds formation means more energy.
Answered by | 03 Mar, 2011, 10:36: AM
Application Videos
Concept Videos
CBSE 12-science - Chemistry
Asked by mallavaramchandrika6 | 29 Sep, 2021, 05:27: PM
CBSE 12-science - Chemistry
Asked by Akshij Nanda | 11 Mar, 2021, 10:21: AM
CBSE 12-science - Chemistry
Asked by sivakapuganti1 | 26 Aug, 2020, 08:57: PM
CBSE 12-science - Chemistry
Asked by spoorthysaienelluri | 23 May, 2020, 11:07: AM
CBSE 12-science - Chemistry
Asked by shreemuvijihari | 18 May, 2020, 06:29: PM
CBSE 12-science - Chemistry
Asked by vermahitesh124 | 12 May, 2020, 07:58: AM
CBSE 12-science - Chemistry
Asked by yogeshsulakh | 07 Feb, 2020, 09:28: AM
CBSE 12-science - Chemistry
Asked by tlb2bpartner | 21 Aug, 2019, 10:28: PM
CBSE 12-science - Chemistry
Asked by ntg432000 | 22 May, 2019, 08:10: PM
CBSE 12-science - Chemistry
Asked by kripanjalihimansu | 01 Mar, 2019, 04:16: PM