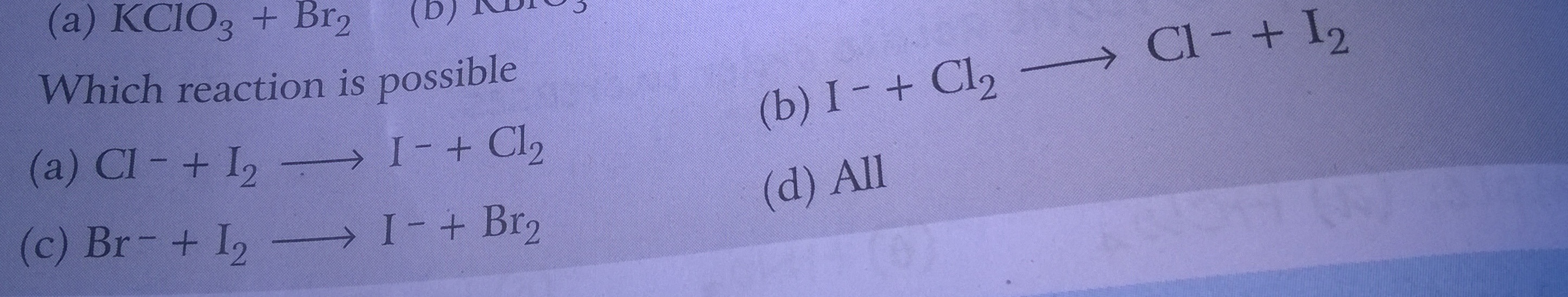CBSE Class 11-science Answered
sir,here is my question on oxidation number
Asked by | 05 Mar, 2008, 02:41: PM
how did the oxidation number of P in HPO2 came to be as +1 in the following redox reaction
P4(s) + OH-(aq) → PH3(g)+ HPO-2 (aq)
Hydrogen and oxygen are +1 and -2 respectively assume P to be x than -1 = +1 + -2X2 + x
 x = +2.
x = +2.
Answered by | 20 Dec, 2017, 04:49: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by jaip83491 | 24 Jan, 2021, 10:43: AM
CBSE 11-science - Chemistry
Asked by sulaikhasulu393 | 07 Jun, 2020, 10:51: PM
CBSE 11-science - Chemistry
Asked by debjit_dm | 04 May, 2020, 03:08: PM
CBSE 11-science - Chemistry
Asked by Balbir | 30 Jun, 2019, 03:45: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 05 Sep, 2018, 01:32: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 09 Aug, 2018, 04:52: PM
CBSE 11-science - Chemistry
Asked by dineshchem108 | 17 Jul, 2018, 06:02: PM
CBSE 11-science - Chemistry
Asked by vaagai2353 | 29 Jun, 2018, 09:55: PM
CBSE 11-science - Chemistry
Asked by Balbir | 19 Jun, 2018, 06:36: PM