CBSE Class 12-science Answered
It's the comparison of energy/unit amount of fissioned or fused, that shows up that more energy is released in fusion than fission.
However you may refer to this detailed explanation available from web,
To answer this you need to look at the binding energy per nucleon graph as follows:
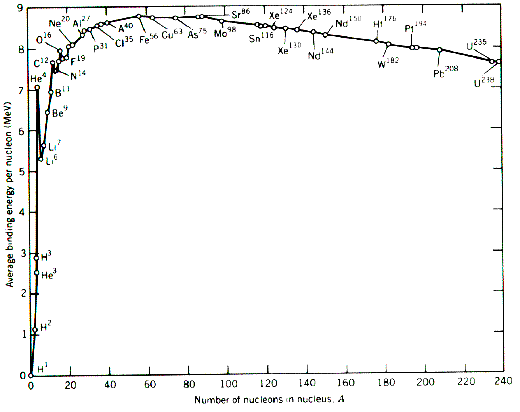
Now lets look at fission. An example of fission is when a Uranium-235 atom is split by a neutron into a Barium-144 atom, a Krypton-89 atom and three neutrons. Now looking at the graph the binding energy per nucleon for Uranium is about 7.6MeV and for Barium around 8.3 giving an increase in binding energy during fission of about 0.7MeV per nucleon, or a total of 164.5MeV in total. In a fusion reaction firstly two hydrogens fuse to form a deuterium (an isotope of hydrogen with nucleon no 2), a positron and an electron neutrino. Then the deuterium fuses with another hydrogen to form Helium-3 and a photon of energy. Finally two Helium-3s fuse forming a Helium nucleus and two hydron nuclei. Considering the mass of the four protons/hydrogen nuclei (4.029106u) and the mass of the Helium produced (4.002603u) we get a mass difference of 0.026503u or 24.69MeV. So it is easy to see that fusion reactions give out more energy per reaction. However, the energy per unit mass is more relevant. This is 0.7MeV for fission and 6.2MeV for fusion so it is obvious that fusion is the more effective nuclear reaction. However, you must remember that an enormous amount of energy is required in order for these reactions to occur at all - that is why fusion is not yet a practical source of energy.
Regards,
Team,
TopperLearning.