CBSE Class 11-science Answered
QUESTION
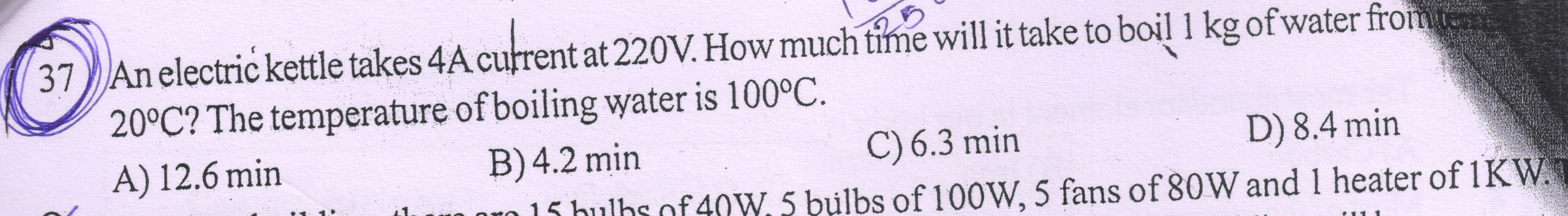
Asked by gpnkumar0 | 02 Oct, 2017, 10:57: AM
The heat supplied to the kettle by the mains is H = VIt
Now, this heat is used to boil the water from 20 °C to 100 °C.
This heat used up is Q = mcΔT
Now, according to principle of calorimetry, H = Q.
VIt = mcΔT
t = (mcΔT)/VI = (1 × 4.2 × 103 × (373 - 293))/(220 × 4) = 381.8 s = 381.8/60 = 6.3 min
Hence, the correct answer is option C.
Answered by Romal Bhansali | 25 Oct, 2017, 03:43: PM
Concept Videos
CBSE 11-science - Physics
Asked by rajualpine16 | 19 Aug, 2020, 07:42: AM
CBSE 11-science - Physics
Asked by hazim.mohaamad2003 | 02 Mar, 2019, 12:10: PM
CBSE 11-science - Physics
Asked by abhishekmishra1031 | 12 Feb, 2019, 03:10: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 17 Apr, 2015, 09:57: AM
CBSE 11-science - Physics
Asked by Topperlearning User | 17 Apr, 2015, 09:56: AM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM