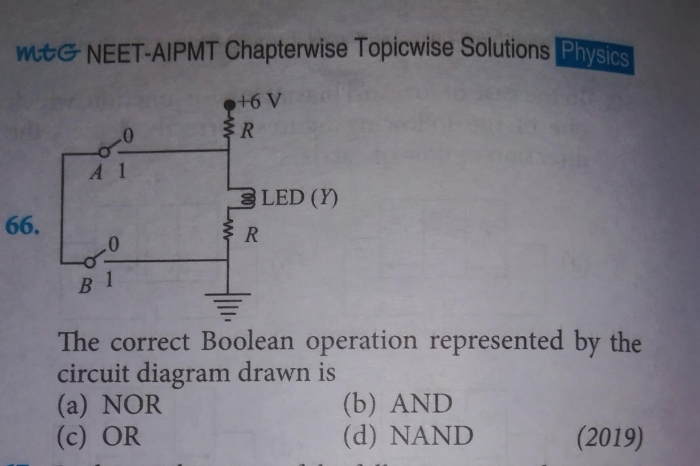
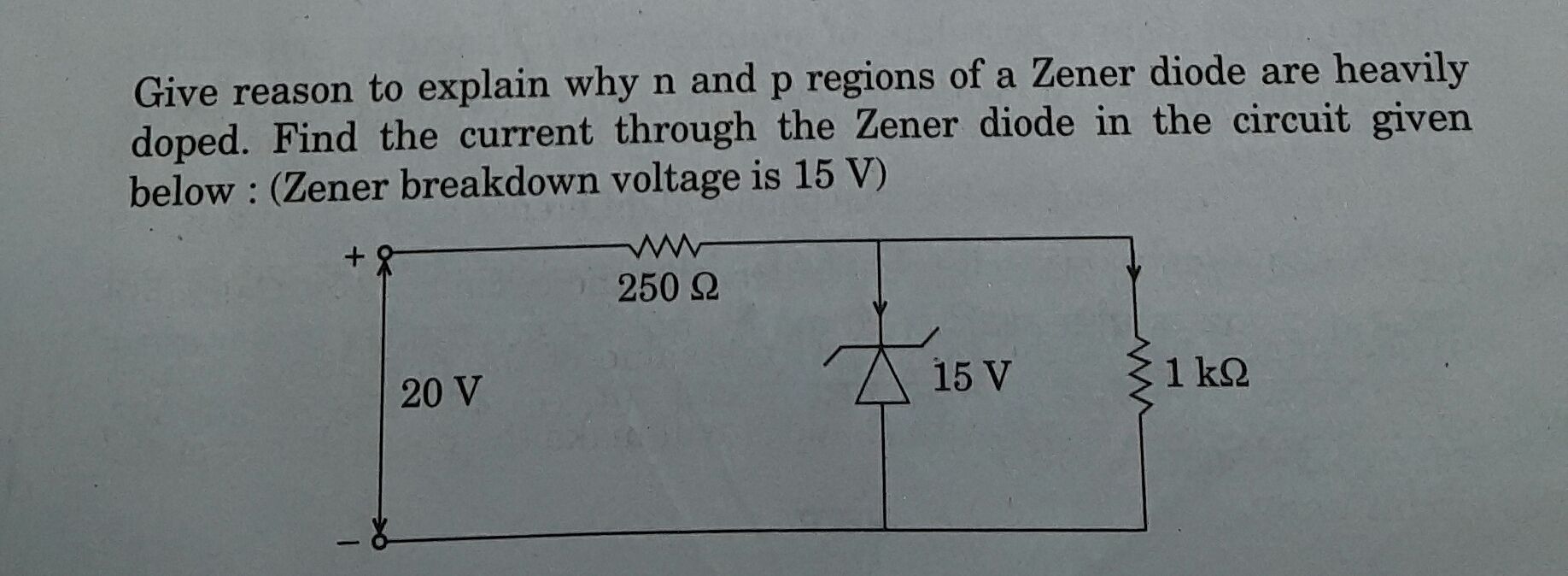
CBSE Class 12-science Answered
We know that there are discrete energy states in a free atom. But in solids as because there are a large number of atoms these available energy states combine to form bands. It is extension of molecular orbitals to bonds between solids.
We all know that valence electrons take part in conduction process and if an electron has only the energy of the valance shell, it would remain attached to the atom and no conduction takes place. This common energy state is the valance band. Now if there be some energy given to the electrons so that they may come out of their valance shells and become free electrons and thereby take part in conduction. This common energy state is known as the conduction band. Between the valance band and the conduction band there is the forbidden gap. In insulators the gap is very large so very very few electrons can reach the conduction band. While in conductors this gap is very very small and conduction band almost overlaps the valance band. Thus lot of electrons are present in the conduction band and conduction takes place. In semi-conductor this gap is lesser than that of insulators but more than that of conductors.