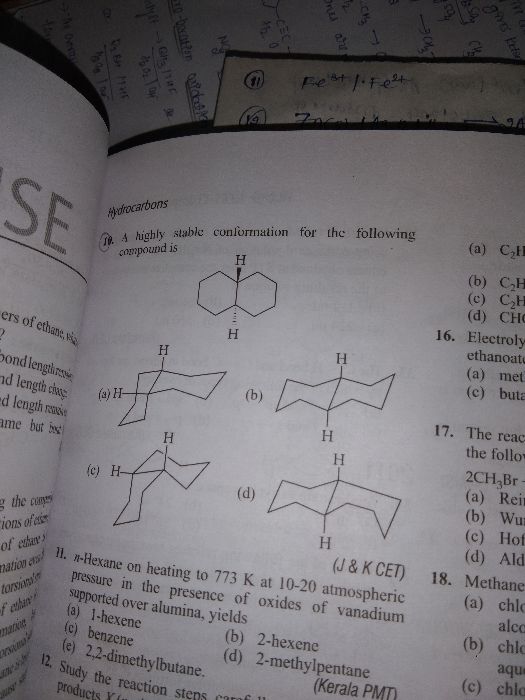
CBSE Class 11-science Answered
Octane number
Octane number denotes the percentage (by volume) of iso-octane (a type of octane) in a combustible mixture (containing iso-octane and normal-heptane) whose 'anti-knocking' characteristics match those of the gas being tested.
Measure of the ignition quality of gas (gasoline or petrol). Higher this number, the less susceptible is the gas to 'knocking' (explosion caused by its premature burning in the combustion chamber) when burnt in a standard (spark-ignition internal combustion) engine.
In the older vehicles, high octane numbers were achieved by adding lead tetraethyl to the gas (the 'leaded gas'), a pollutant that contributes to lead poisoning (see Lead). In the newer vehicles, the same result is achieved by the engine design that increases turbulence in the combustion chamber, and/or by adding aromatic hydrocarbons (such as xylenes) and oxygenates (oxygen-containing compounds such as alcohols) to the gas (the 'unleaded gas'). Also called Octane rating.
Cetane number
Measure of the ignition quality of diesel fuel; higher this number, the easier it is to start a standard (direct-injection) diesel engine. It denotes the percentage (by volume) of cetane (chemical name Hexadecane) in a combustible mixture (containing cetane and 1-methylnapthalene) whose ignition characteristics match those of the diesel fuel being tested.