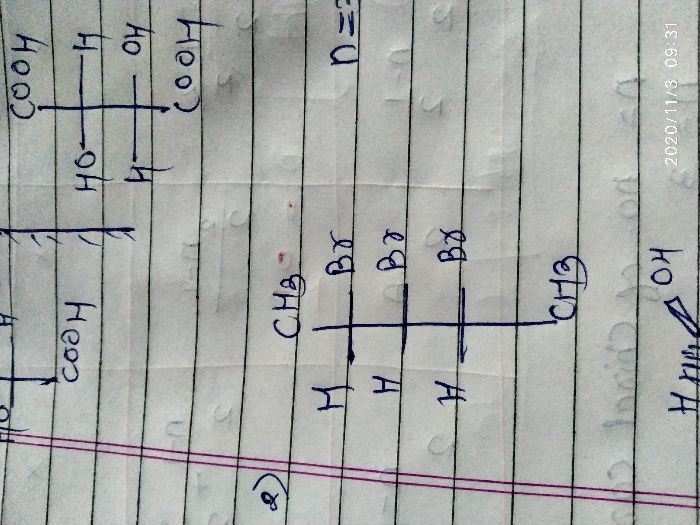CBSE Class 11-science Answered
please explain TAUTOMERISM.
please provide me with some examples of it .
Asked by preethambasilmartis | 23 Jun, 2016, 08:12: AM
Tautomers are isomers of organic compounds that readily interconvert by a chemical reaction called tautomerization. Commonly this reaction results in the formal migration of a hydrogen atom or proton, accompanied by a switch of a single bond and adjacentdouble bond. In solutions where tautomerization is possible, a chemical equilibrium of the tautomers will be reached. The exact ratio of the tautomers depends on several factors, including temperature, solvent, and pH. The concept of tautomers that are interconvertible by tautomerizations is called tautomerism. Tautomerism is a special case of structural isomerism and can play an important role in non-canonical base pairing in DNA and especially RNA molecules.
Answered by Vaibhav Chavan | 23 Jun, 2016, 04:28: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by ap4450962 | 12 Mar, 2024, 07:35: PM
CBSE 11-science - Chemistry
Asked by shahintkjnv2016 | 13 Jun, 2022, 07:17: PM
CBSE 11-science - Chemistry
Asked by rohus442 | 03 Nov, 2020, 09:31: AM
CBSE 11-science - Chemistry
Asked by ashok.amireddi | 01 May, 2020, 10:33: PM
CBSE 11-science - Chemistry
Asked by mufeedatvp2000 | 13 Apr, 2020, 08:36: PM
CBSE 11-science - Chemistry
Asked by prakriti12oct | 12 Dec, 2019, 12:00: AM
CBSE 11-science - Chemistry
Asked by ajaysankhala051 | 02 Sep, 2019, 02:26: PM
CBSE 11-science - Chemistry
Asked by musira29rahman | 30 Aug, 2019, 05:09: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 27 Aug, 2019, 06:33: PM
CBSE 11-science - Chemistry
Asked by ajaysankhala051 | 26 Aug, 2019, 09:39: PM