CBSE Class 11-science Answered
pl experts ans as soon as possible
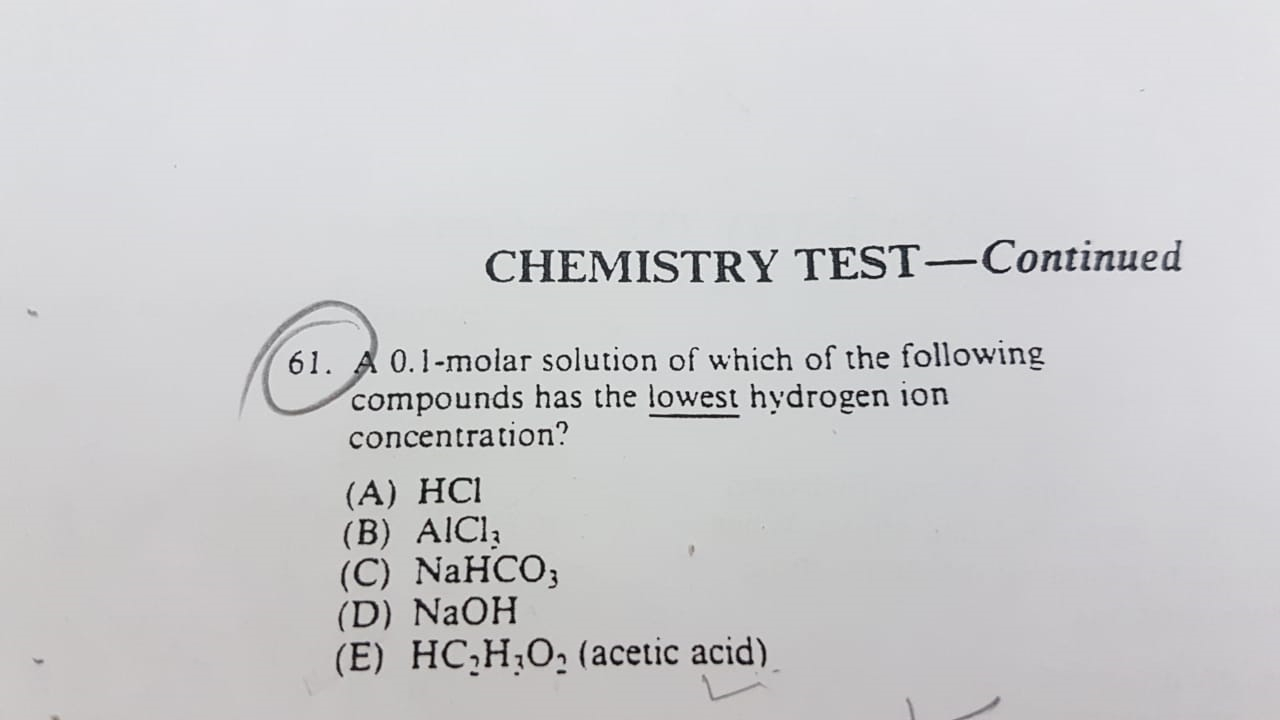
Asked by jain.pradeep | 14 Apr, 2019, 12:34: AM
(A) HCl- Strong acid.
(B) AlCl3 - Salt of a strong acid and weak base
(C) NaHCO3 - Salt of a weak acid and strong base
(D) NaOH- Strong base
(E) CH3COOH- weak acid
When concentration is the same, Strong base have the lowest number of hydrogen ions in aqueous solution.
Answered by Ravi | 15 Apr, 2019, 03:36: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by sarojlaxmiacharjya | 03 Jan, 2022, 08:50: PM
CBSE 11-science - Chemistry
Asked by cjam41665 | 09 Oct, 2021, 11:11: PM
CBSE 11-science - Chemistry
Asked by rishika62124 | 03 Mar, 2021, 05:02: AM
CBSE 11-science - Chemistry
Asked by jyotijhajharia39 | 06 Jan, 2021, 11:41: PM
CBSE 11-science - Chemistry
Asked by nsaikumar33 | 15 Aug, 2020, 11:50: AM
CBSE 11-science - Chemistry
Asked by swati2678 | 10 Aug, 2020, 01:58: PM
CBSE 11-science - Chemistry
Asked by achamerahul2 | 17 Apr, 2020, 10:50: AM
CBSE 11-science - Chemistry
Asked by achamerahul2 | 17 Apr, 2020, 10:44: AM
CBSE 11-science - Chemistry
Asked by achamerahul2 | 14 Apr, 2020, 02:42: PM
CBSE 11-science - Chemistry
Asked by SanskarAgarwal86 | 29 Feb, 2020, 04:36: AM