CBSE Class 12-science Answered
Osmotic Pressure of 0.4% urea solution is 1.64 atm and that of 3.42% cane sugar is 2.46 atm. When the above two solutions are mixed , the OP of the solution is
(a) 0.82 atm
(b) 2.46 atm
(c) 1.64 atm
(d) 4.10 atm
Asked by Balbir | 27 Jul, 2019, 05:02: PM
When two solutions are mixed.
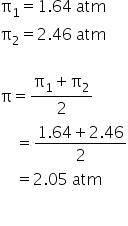
Answered by Ravi | 27 Jul, 2019, 09:19: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by rashmij34 | 27 Feb, 2024, 04:42: PM
CBSE 12-science - Chemistry
Asked by premkhare2006 | 24 Jan, 2024, 09:50: AM
CBSE 12-science - Chemistry
Asked by kaushikmisty07 | 31 Dec, 2023, 11:42: AM
CBSE 12-science - Chemistry
Asked by KRISHPATEL.soc | 21 Jun, 2021, 05:58: PM
CBSE 12-science - Chemistry
Asked by dhrubajyoti.das | 09 May, 2021, 09:54: PM
CBSE 12-science - Chemistry
Asked by tiwariaatman | 31 Jul, 2020, 05:10: PM
CBSE 12-science - Chemistry
Asked by yogendrasoni142 | 08 Jun, 2020, 05:43: PM
CBSE 12-science - Chemistry
Asked by santosh357m | 28 Apr, 2020, 09:28: AM
CBSE 12-science - Chemistry
Asked by Balbir | 27 Jul, 2019, 05:02: PM
CBSE 12-science - Chemistry
Asked by ajaysankhala051 | 04 Jun, 2019, 02:28: PM






