CBSE Class 10 Answered
Na has atomic no. 11 whereas K has atomic no. 19 then why patassium's name comes first in reactivity series?
Asked by Kalita Padmanath | 30 Jun, 2014, 10:57: AM
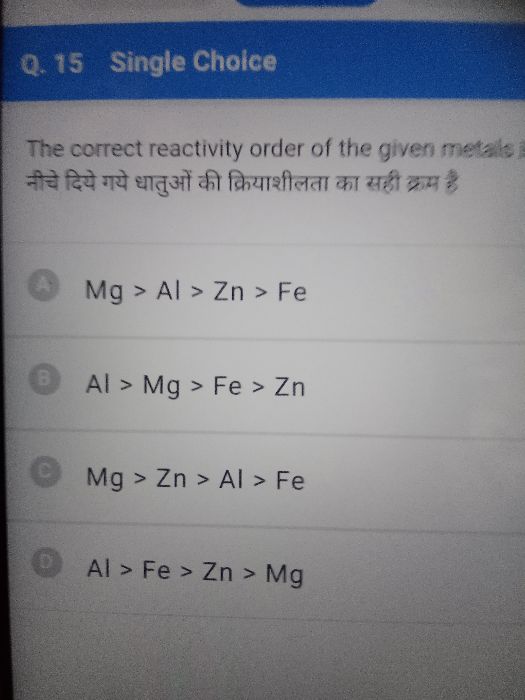
The arrangement of metals in the decreasing order of their reactivity is called the activity or reactivity series of the metals.
The metal potassium is kept at the top of the series because it is most reactive.
Also it is most electropositive as it oxidises i.e., gives up valence electrons.
Answered by Vaibhav Chavan | 30 Jun, 2014, 11:14: AM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by ritik9897022 | 05 Feb, 2024, 09:42: PM
CBSE 10 - Chemistry
Asked by shubham.sharma80634 | 05 Oct, 2021, 09:18: AM
CBSE 10 - Chemistry
Asked by virkarman36 | 08 Aug, 2021, 09:24: AM
CBSE 10 - Chemistry
Asked by dnupadhyay214 | 13 Mar, 2021, 12:01: PM
CBSE 10 - Chemistry
Asked by Vishavjet567 | 31 Oct, 2020, 10:52: AM
CBSE 10 - Chemistry
Asked by aryanluniwal1516 | 12 Sep, 2020, 11:43: AM
CBSE 10 - Chemistry
Asked by broprint18 | 07 Jun, 2020, 04:16: PM
CBSE 10 - Chemistry
Asked by prakharsingh167 | 25 May, 2020, 10:20: PM
CBSE 10 - Chemistry
Asked by sonaliagarwal172 | 16 May, 2020, 10:16: AM