CBSE Class 12-science Answered
Mole fraction of glycerine in a solution of 36g of water and 46 g of glycerine
Asked by suryanshmishr | 05 Jul, 2019, 12:30: PM
Given:
Mass of water = 36gm
Mass of glycerine = 46 gm
Molar mass of watet= 18
Molar mass of glycerine= 92
No. of moles of water=
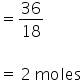
No. of moles of glycerine =
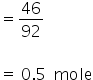
Total moles= 2+0.5
= 2.5 moles
Mole fraction of glycerine =

Mole fraction of water
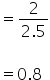
Answered by Varsha | 07 Jul, 2019, 12:28: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by hannamaryphilip | 17 Apr, 2024, 11:20: PM
CBSE 12-science - Chemistry
Asked by sameerteli003 | 08 Apr, 2024, 11:48: PM
CBSE 12-science - Chemistry
Asked by saritanohar22 | 13 Jan, 2024, 01:25: PM
CBSE 12-science - Chemistry
Asked by kamlesh.kumar.malee | 20 Dec, 2023, 06:59: AM
CBSE 12-science - Chemistry
Asked by shamiyaali732 | 26 Sep, 2023, 02:00: AM
CBSE 12-science - Chemistry
Asked by elabarman58 | 23 Jan, 2023, 09:39: AM
CBSE 12-science - Chemistry
Asked by gauravrastogi577 | 16 Aug, 2022, 06:18: PM
CBSE 12-science - Chemistry
Asked by gauravrastogi577 | 16 Aug, 2022, 06:17: PM
CBSE 12-science - Chemistry
Asked by shiv.pama83 | 27 Nov, 2021, 05:50: AM
CBSE 12-science - Chemistry
Asked by mdarsadazizh | 31 Aug, 2021, 01:02: PM







