ICSE Class 8 Answered
Valency:
Valency is the combining capacity of an element or radical.
For example, the valency of carbon is 4 because it combines with four atoms of hydrogen to yield methane (CH4).
Variable Valency
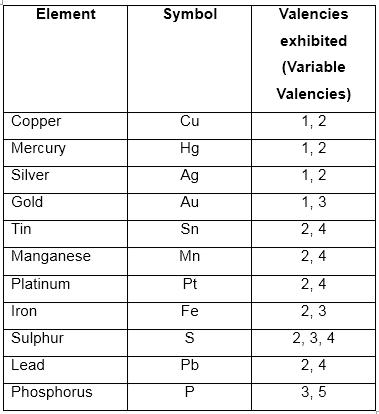
Sometimes, the same element may exhibit one valency in one compound and another valency in other compound. This property is called variable valency.
Examples of variable valencies:
Ions or Radicals
An ion or radical is an atom or a group of atoms of the same or different elements which behave as a single unit with a positive or negative charge on it.
Radicals have their own combining power based on which they form chemical formulae.
Types:
Radicals are of two types:
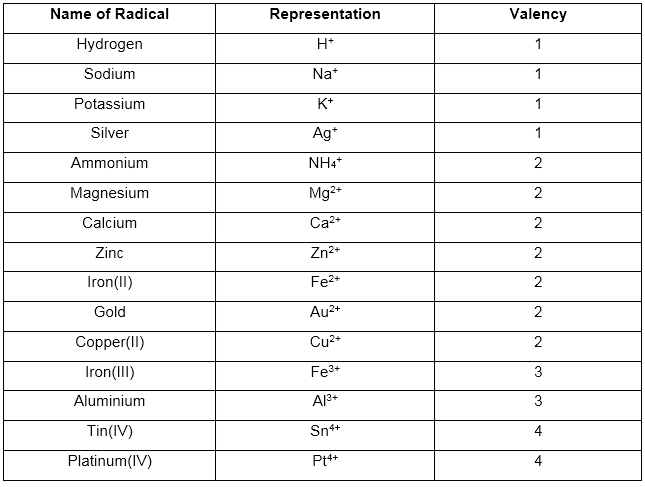
(1) Basic or positive radicals:
These have a positive charge and are also called cations.
Example: All metallic ions and ammonium ion (NH4+) are basic radicals.
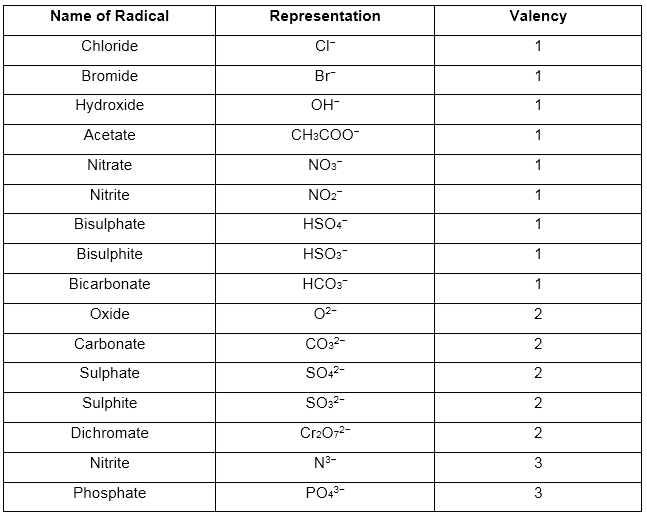
(2) Acid or negative radicals:
These have a negative charge and are also called anions.
Example: Most of all non-metallic ions and groups of non-metallic atoms with a negative charge are acid radicals.
Representation of some positive radicals and their valency:
Representation of some negative radicals and their valency:
You have to remember the valency and variable valencies of these elements and radicals.