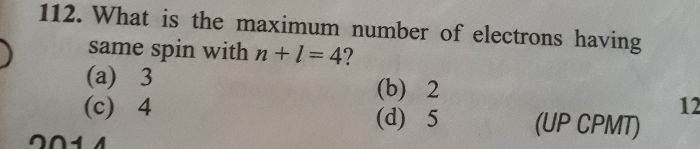CBSE Class 11-science Answered
in what wau heisenberg uncertainity principle contradicts the concept of stationary orbit
for electron as sugested by bohr? a electron has a speed of 500 m/s witha
an uncertainity of0.02% what is the uncertainity in locating the position ?
given h= 6.6x10 tothe power minus 34 m-2s-1
which state of the triply ionised Be3+ has the same orbital radius as that of the ground state of hydrogen?
compare the energies of the two states.
Asked by patidarshm | 11 Dec, 2015, 12:05: PM
Dear patidarshm@gmail.com
Thanks for asking us a question in Ask the Expert section of TopperLearning.com.
In case of multiple questions within a query, please post each question individually and let us know where you are getting stuck so that we would be able to explain things better.
Solution for your first query,
Regards
Topperlearning Team.
Topperlearning Team.
Answered by Prachi Sawant | 13 Dec, 2015, 12:54: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by deba.biswas561 | 14 Jun, 2022, 08:07: AM
CBSE 11-science - Chemistry
Asked by Neetukhaiwal | 24 Jul, 2021, 11:18: AM
CBSE 11-science - Chemistry
Asked by salvadormiranda2509 | 13 Feb, 2021, 12:53: PM
CBSE 11-science - Chemistry
Asked by sulaikhasulu393 | 01 May, 2020, 04:37: PM
CBSE 11-science - Chemistry
Asked by prashantyadav592 | 29 Jan, 2020, 09:39: AM
CBSE 11-science - Chemistry
Asked by Debdulal | 29 Aug, 2019, 04:47: PM
CBSE 11-science - Chemistry
Asked by sairamsribaba80 | 28 Aug, 2019, 06:54: PM
CBSE 11-science - Chemistry
Asked by timeyashkadam | 19 Apr, 2019, 10:22: AM
CBSE 11-science - Chemistry
Asked by himankarbhati3 | 21 Sep, 2018, 08:29: AM
CBSE 11-science - Chemistry
Asked by ramandeep.kaur | 06 Sep, 2018, 01:21: PM