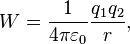CBSE Class 12-science Answered
The electrostatic potential energy is the energy of an electrically charged particle (at rest) in an electric field. It is defined as the work that must be done to move it from an infinite distance away to its present location, in the absence of any non-electrical forces on the object. This energy is non-zero if there is another electrically charged object nearby.
The simplest example is the case of two point-like objects A1 and A2 with electrical chargesq1 and q2. The work W required to move A1 from an infinite distance to a distance r away from A2 is given by:
where ?0 is the electric constant.
This equation is obtained by integrating the coulomb force between the limits of infinity and r.
A related quantity called electric potential (commonly denoted with a V for voltage) is equal to the electric potential energy per unit charge. So as we move further potential energy will decrease.
- The electron is held in a circular orbit by electrostatic attraction. The centripental force is equal to the coulomb force.
- where me is the mass, e is the charge of the electron and ke is coulomb's constant. This determines the speed at any radius:
- It also determines the total energy at any radius:
- The total energy is negative and inversely proportional to r. This means that it takes energy to pull the orbiting electron away from the proton. For infinite values of r, the energy is zero, corresponding to a motionless electron infinitely far from the proton.
- Therefore, as we move away from the nucleus,kinetic energy decreases.