CBSE Class 10 Answered
In a period(including Noble gases)which element has the largest size? Explain by giving example with the reason.
Asked by shiphalijain | 10 Mar, 2017, 08:06: PM
Atomic radius goes on decreasing along the period from left to right as electron goes on to the same shell and the attraction between positive nucleus and outer shell negatively charged electrons goes on increasing from left to right across a period.
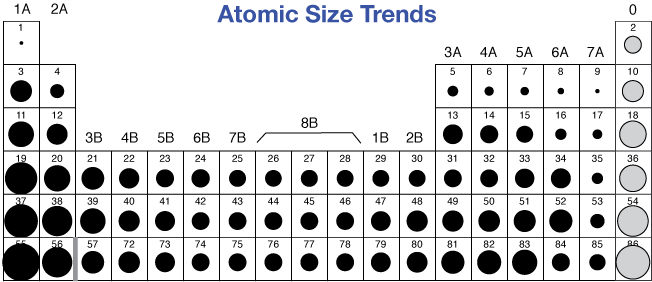
Answered by Prachi Sawant | 12 Mar, 2017, 06:27: PM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by dhumalchhaya13 | 26 May, 2022, 11:05: PM
CBSE 10 - Chemistry
Asked by kanchanbalpande82 | 12 Apr, 2022, 11:01: AM
CBSE 10 - Chemistry
Asked by kanchanbalpande82 | 12 Apr, 2022, 11:01: AM
CBSE 10 - Chemistry
Asked by waghmaresheetal78 | 27 Dec, 2021, 05:02: PM
CBSE 10 - Chemistry
Asked by ksheera36 | 03 Jun, 2021, 08:35: PM
CBSE 10 - Chemistry
Asked by vungtsaniyanthan | 16 May, 2021, 06:32: PM
CBSE 10 - Chemistry
Asked by sinhagopalakumara | 01 May, 2021, 08:16: PM
CBSE 10 - Chemistry
Asked by advssdrall | 26 Mar, 2021, 07:43: AM
CBSE 10 - Chemistry
Asked by shettyshrinidhi271 | 07 Jan, 2021, 05:04: PM
CBSE 10 - Chemistry
Asked by adipadmakarri | 05 Dec, 2020, 06:41: PM










