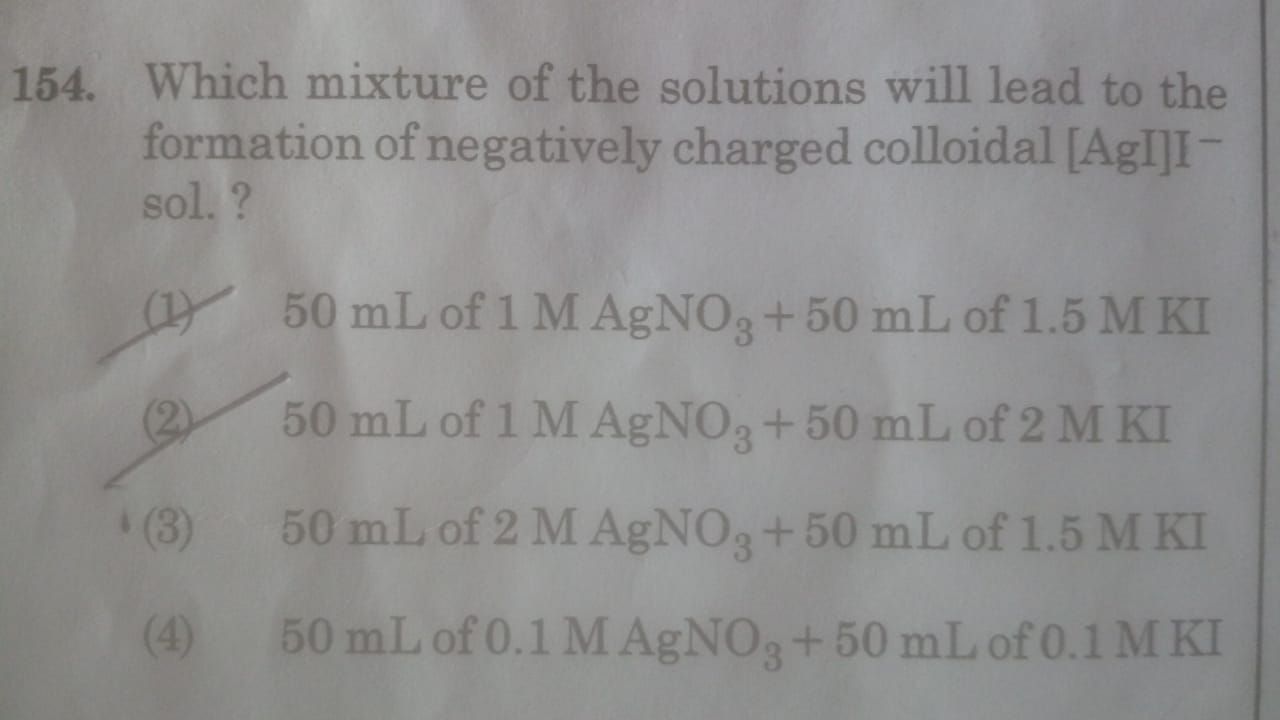CBSE Class 12-science Answered
Illustrate graphically the effect of temperature on adsorption at constant pressure both for physisorption and chemisorption.besides frundlich's and Langmuir's adsorption isotherms what other such isotherms are there?discuss briefly on them.
Asked by Chandropal Parashor | 05 Sep, 2014, 06:30: AM
An increase in temperature decreases physical adsorption whereas, chemisorption first increases and then decreases.
There are three main adsorption isotherms:
- Frundlich's adsorption isotherm
- Langmuir's adsorption isotherm
- BET adsorption Isotherm
Other than above isotherms there are some models Polanyi Potential Theory and Dubinin-Radushkevich Equation which are for microporous adsorption.
Answered by Arvind Diwale | 05 Sep, 2014, 11:00: AM
Application Videos
Concept Videos
CBSE 12-science - Chemistry
Asked by mallavaramchandrika6 | 29 Sep, 2021, 05:27: PM
CBSE 12-science - Chemistry
Asked by Akshij Nanda | 11 Mar, 2021, 10:21: AM
CBSE 12-science - Chemistry
Asked by sivakapuganti1 | 26 Aug, 2020, 08:57: PM
CBSE 12-science - Chemistry
Asked by spoorthysaienelluri | 23 May, 2020, 11:07: AM
CBSE 12-science - Chemistry
Asked by shreemuvijihari | 18 May, 2020, 06:29: PM
CBSE 12-science - Chemistry
Asked by vermahitesh124 | 12 May, 2020, 07:58: AM
CBSE 12-science - Chemistry
Asked by yogeshsulakh | 07 Feb, 2020, 09:28: AM
CBSE 12-science - Chemistry
Asked by tlb2bpartner | 21 Aug, 2019, 10:28: PM
CBSE 12-science - Chemistry
Asked by ntg432000 | 22 May, 2019, 08:10: PM
CBSE 12-science - Chemistry
Asked by kripanjalihimansu | 01 Mar, 2019, 04:16: PM