CBSE Class 12-science Answered
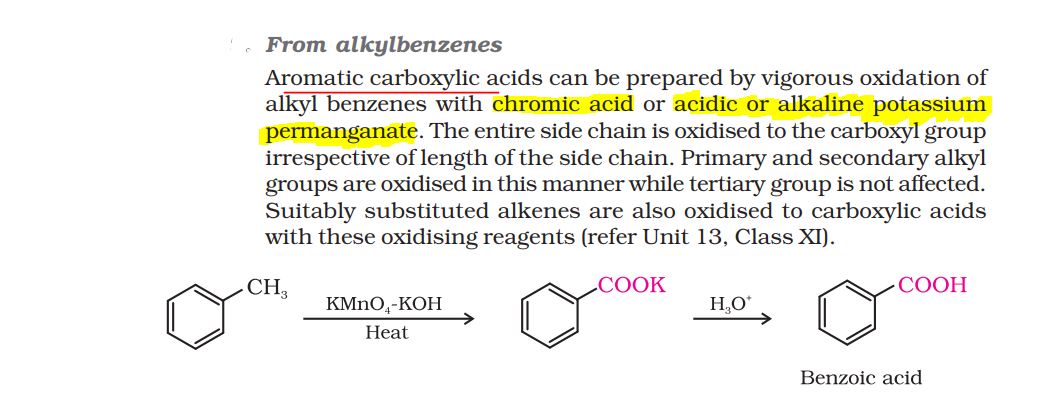
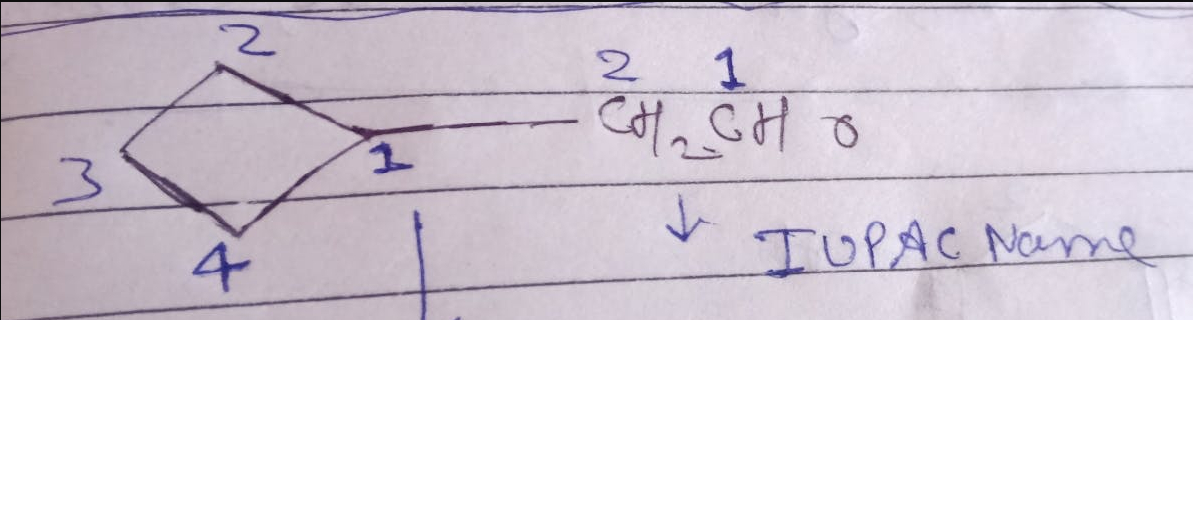
If in a reaction ,that is on oxidation A gives two carboxylic acids B and C where B is ethanoic acid and C is butanoic acid , how can we come to the conclusion that the keto group is present on the second carbon?
Asked by sarbsukhmani | 12 Aug, 2010, 12:00: AM
becaue one of the acids formed is ethanoic acid which has two carbon atoms.
Secondly, according to the rule the C=O group stays with the alkyl group which has lower number of carbon atoms.
Answered by | 12 Aug, 2010, 04:03: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by ukg8612 | 15 Apr, 2024, 07:36: PM
CBSE 12-science - Chemistry
Asked by ajayarchi | 08 Feb, 2024, 03:43: AM
CBSE 12-science - Chemistry
Asked by pallasriramulu9 | 24 Dec, 2023, 06:05: AM
CBSE 12-science - Chemistry
Asked by bsaheliya | 22 Dec, 2023, 09:53: PM
CBSE 12-science - Chemistry
Asked by ygarg8323 | 18 Apr, 2022, 12:47: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 30 Jun, 2021, 04:52: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 29 Jun, 2021, 08:36: AM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 28 Jun, 2021, 02:34: PM
CBSE 12-science - Chemistry
Asked by saimerala007 | 22 May, 2021, 02:08: PM
CBSE 12-science - Chemistry
Asked by amitkumar.cis | 31 Dec, 2020, 10:45: AM