CBSE Class 12-science Answered
If 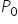 and
and 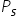 are the vapour pressures of the solvent and the solution respectively and
are the vapour pressures of the solvent and the solution respectively and 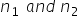 are the mole fractions of the solvent and solutions respectiively, then:
are the mole fractions of the solvent and solutions respectiively, then:
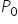 and
and 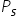 are the vapour pressures of the solvent and the solution respectively and
are the vapour pressures of the solvent and the solution respectively and 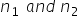 are the mole fractions of the solvent and solutions respectiively, then:
are the mole fractions of the solvent and solutions respectiively, then: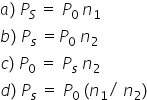
Asked by ayushi | 16 May, 2016, 10:56: AM
Answer is (a).
Answered by Arvind Diwale | 16 May, 2016, 12:01: PM
CBSE 12-science - Chemistry
Asked by sagarmishra | 27 Feb, 2024, 04:01: PM
CBSE 12-science - Chemistry
Asked by ayazanwarneet | 03 Jan, 2023, 10:58: PM
CBSE 12-science - Chemistry
Asked by gopirammeghwal78 | 05 Aug, 2021, 08:18: AM
CBSE 12-science - Chemistry
Asked by msambyal307 | 30 May, 2021, 02:03: PM
CBSE 12-science - Chemistry
Asked by nishchaymakhija115 | 11 Sep, 2019, 02:31: PM
CBSE 12-science - Chemistry
Asked by pandeyn1604 | 29 May, 2019, 09:16: AM
CBSE 12-science - Chemistry
Asked by rakeshraghav33 | 18 Sep, 2018, 04:22: PM
CBSE 12-science - Chemistry
Asked by Avadhut Katkar | 14 Sep, 2018, 12:37: PM
CBSE 12-science - Chemistry
Asked by minipkda | 21 May, 2018, 10:19: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 20 Jun, 2016, 04:00: PM

