CBSE Class 12-science Answered
I want make a model:3d structure of nacl and simple cube tetrahedral and octahedral void how to make 3d model of this I m in confusion that which diagram is best to make
file: brief explanations about type of solid structure details of nacl,cscl,zns,cafe, antifluoride with diagram
so please tell me fast I start making a model and also tell that which materials should be use for making a model
Asked by Seema Jain | 31 May, 2015, 03:34: PM
Dear Seema,
Thanks for asking us a question in Ask the Expert section of TopperLearning.com.
In case of multiple questions within a query, please post each question individually and let us know where you are getting stuck so that we would be able to explain things better.
Solution for your first query,
You can make 4 x 4 x 4 model of NaCl as given below:
Concept Videos
CBSE 12-science - Chemistry
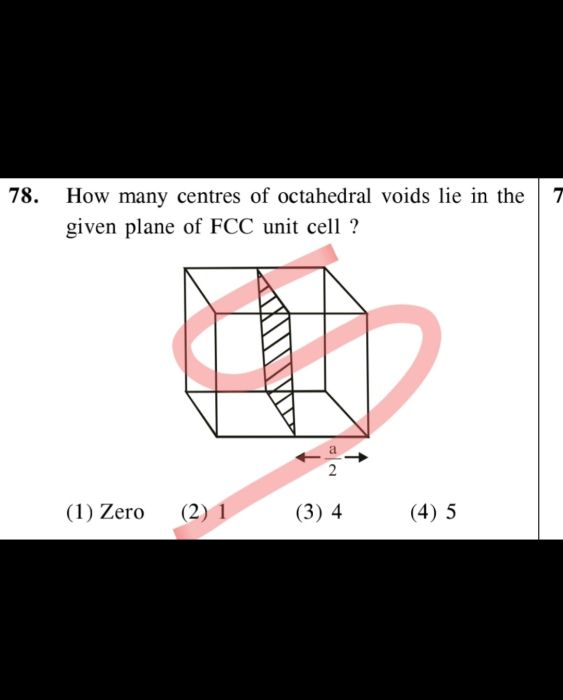
Asked by harshul2019 | 25 May, 2022, 08:52: PM
CBSE 12-science - Chemistry
Asked by arushidabhade | 17 Mar, 2021, 01:24: PM
CBSE 12-science - Chemistry
Asked by kasthurikalvi | 16 Sep, 2020, 03:46: PM
CBSE 12-science - Chemistry
Asked by sulaikhasulu393 | 01 Jul, 2020, 10:08: PM
CBSE 12-science - Chemistry
Asked by anukritisingh8103.bmps | 18 May, 2020, 03:00: PM
CBSE 12-science - Chemistry
Asked by rohitraman1115 | 10 Sep, 2019, 07:10: PM
CBSE 12-science - Chemistry
Asked by prakriti12oct | 05 Aug, 2019, 12:08: AM
CBSE 12-science - Chemistry
Asked by prakriti12oct | 04 Aug, 2019, 09:06: PM
CBSE 12-science - Chemistry
Asked by ranasingh04082002 | 31 Jul, 2019, 02:04: PM
CBSE 12-science - Chemistry
Asked by Saransekar407 | 11 Mar, 2019, 06:51: PM