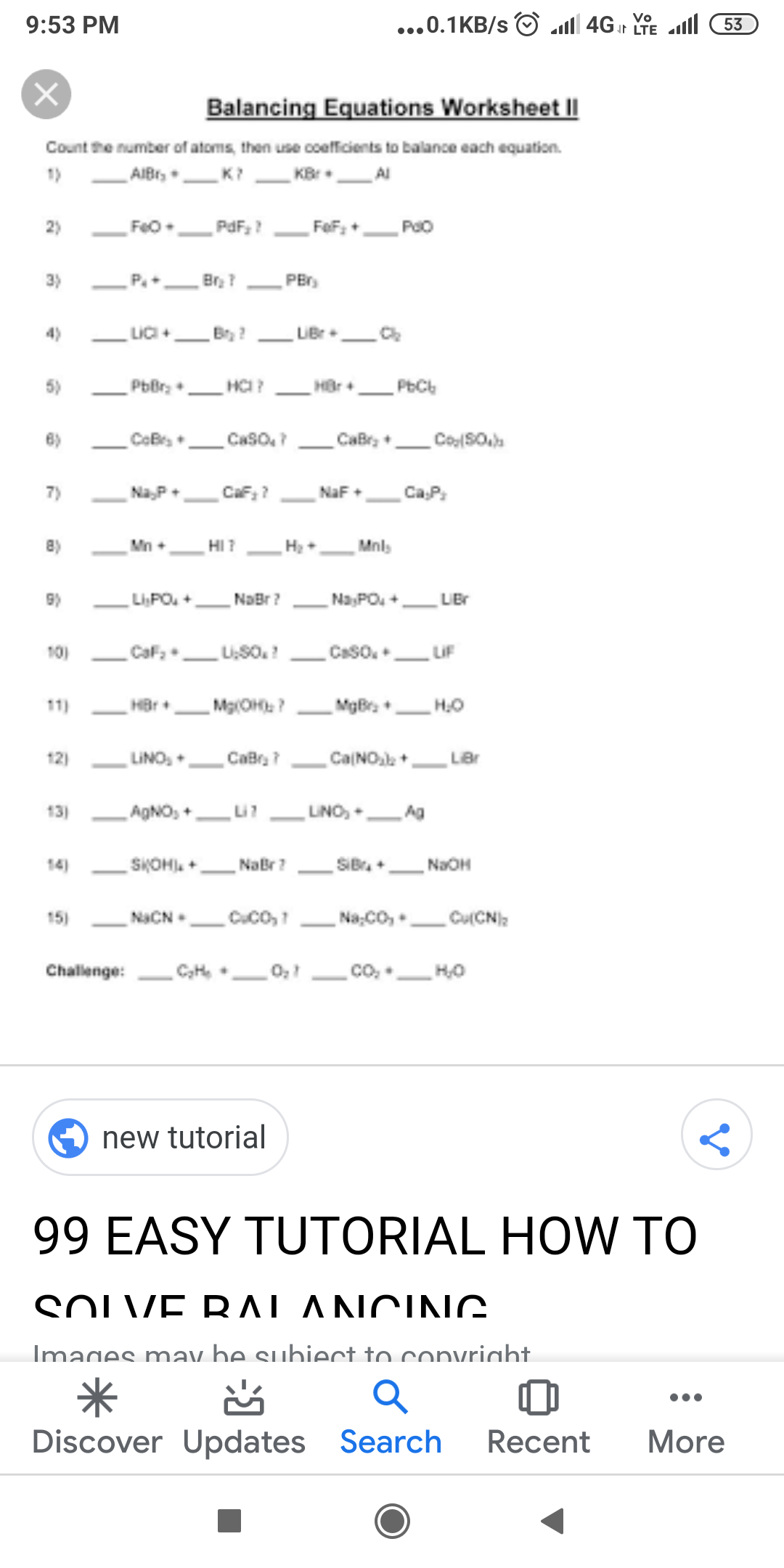
ICSE Class 7 Answered
- Balancing an equation
- In a balanced chemical equation, the total number of atoms of each element in the reactants on the left-hand side of the equation is the same as the number of atoms in the products formed on the right-hand side of the equation.
- The total mass of the reactants is equal to the total mass of the products. The number of atoms of each element before the reaction and after the reaction is equal.
Steps Involved in Balancing a Chemical Equation
Consider the chemical reaction between magnesium and oxygen to understand the steps involved in balancing a chemical equation.
Step 1
Let us first write the word equation for this reaction.
Magnesium + Oxygen → Magnesium oxide
Step 2
Write the chemical equation for the reaction between magnesium and oxygen.
Mg + O2 → MgO
Step 3
Count the number of times an element occurs on both LHS and RHS in this equation.
Mg + O2 → MgO
|
Component |
Reactant |
Product |
|
Magnesium |
1 |
1 |
|
Oxygen |
2 |
1 |
This is an unbalanced equation.
Step 4
- To balance a chemical equation, first draw boxes around each formula. Do not change anything inside the boxes while balancing the equation.
- Choose a reactant or product which has the maximum number of atoms in it. In that compound, select the element which has the maximum number of atoms. In this equation, we will select MgO, i.e. magnesium oxide and the element oxygen in it.
- To balance the oxygen atoms, let us multiply the magnesium oxide molecule by 2 on the right-hand side.
The equation can now be expressed as
|
Component |
Reactant |
Product |
|
Magnesium |
1 |
1 × 2 = 2 |
|
Oxygen |
2 |
1 × 2 = 2 |
Step 5
There are two oxygen atoms on either side of the equation, but one magnesium atom on the reactant's side and two on the product's side. Therefore, multiply the magnesium atom by 2 on the left-hand side.
|
Component |
Reactant |
Product |
|
Magnesium |
1 × 2 = 2 |
2 |
|
Oxygen |
2 |
2 |
The balanced equation is
\Number of atoms of each element of the reactants = Number of atoms of each element of the products
Step 6
Writing specific conditions on the arrow
The reaction is carried out in the presence of ‘heat’. On heating, magnesium combines with oxygen present in the air to form magnesium oxide.
Step 7
Writing symbols of the physical state
- To make a chemical equation more informative, the physical states of the reactants and products are mentioned along with their chemical formulae.
- The gaseous, liquid, aqueous and solid states of the reactants and products are represented by the notations (g), (l), (aq) and (s), respectively.
Using these steps, you can balance any chemical equation.