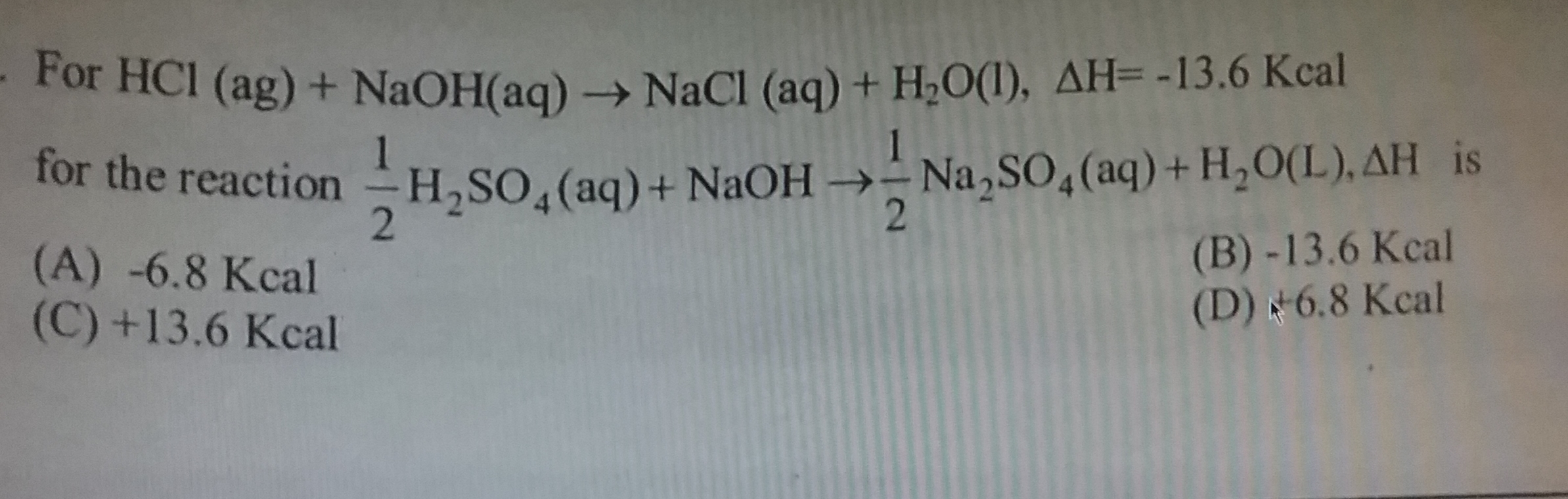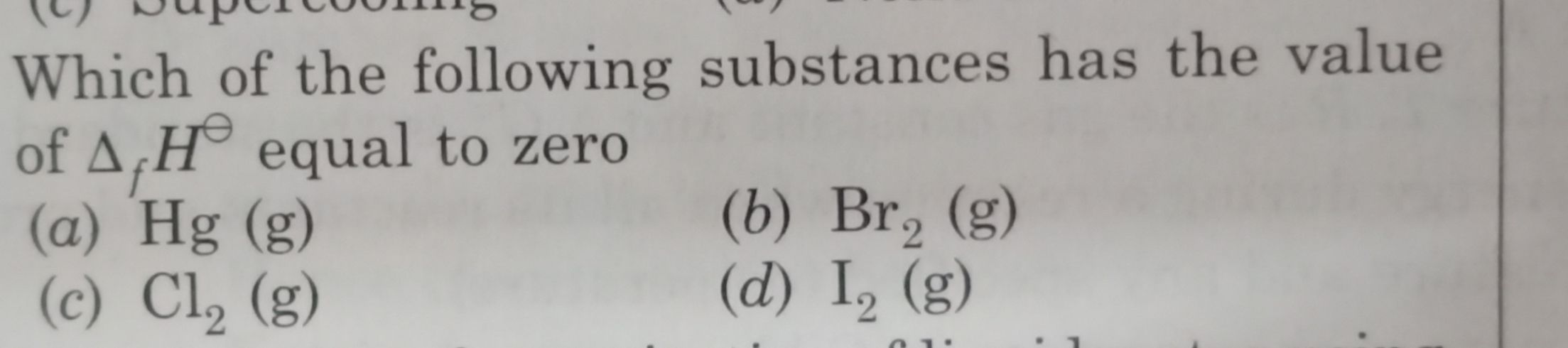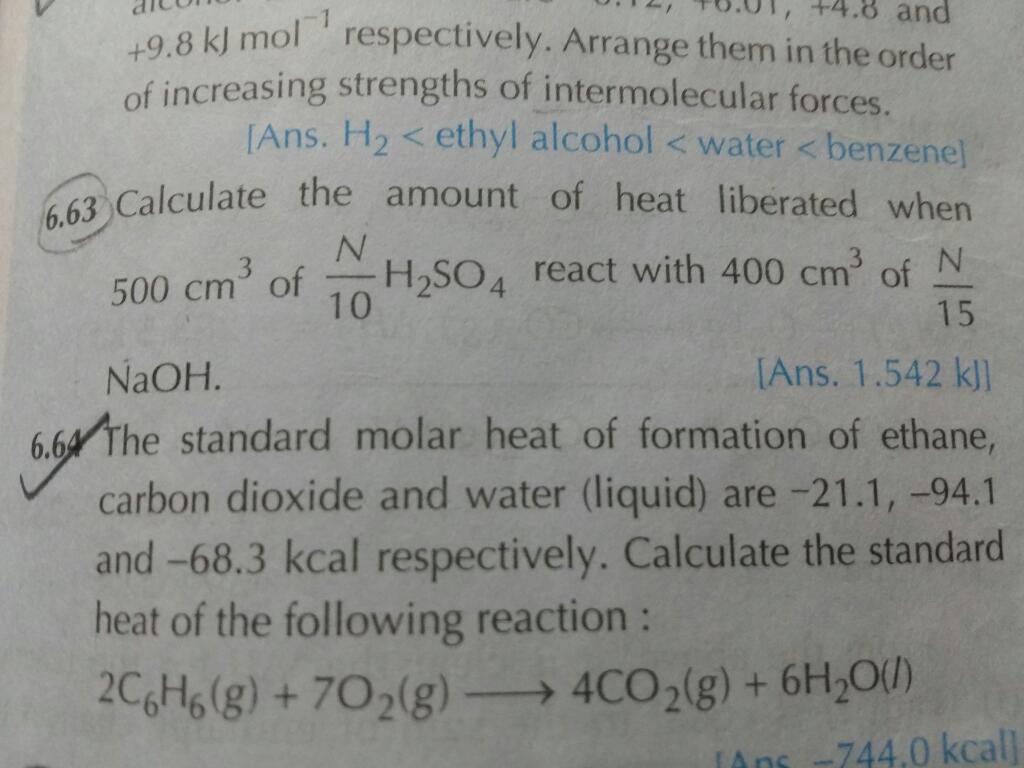CBSE Class 11-science Answered
How is isothermal irreversible expansion cyclic and how is delta H=0 for the process?
Asked by prakriti12oct | 26 Sep, 2019, 01:40: AM
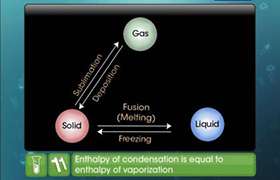
The isothermal irreversible expansion process is an acyclic process because when the gas expands irreversibly then it can not return into its previous state so it is not cyclic. Enthalpy change is zero in the isothermal process because enthalpy is a function of temperature. So when the temperature is constant it is also constant and Enthalpy change=0.
Answered by Ravi | 26 Sep, 2019, 06:17: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by advssdrall | 11 Jan, 2022, 07:44: PM
CBSE 11-science - Chemistry
Asked by adityasolanki7773 | 22 Oct, 2020, 03:40: PM
CBSE 11-science - Chemistry
Asked by pranavisrihari | 08 Sep, 2020, 05:24: PM
CBSE 11-science - Chemistry
Asked by varakalasuchi3 | 28 Mar, 2020, 04:47: PM
CBSE 11-science - Chemistry
Asked by patra04011965 | 09 Nov, 2019, 12:18: PM
CBSE 11-science - Chemistry
Asked by prakriti12oct | 27 Sep, 2019, 01:43: AM
CBSE 11-science - Chemistry
Asked by prakriti12oct | 26 Sep, 2019, 01:40: AM
CBSE 11-science - Chemistry
Asked by sayantan.chem2 | 06 Aug, 2019, 05:07: PM
CBSE 11-science - Chemistry
Asked by lovemaan5500 | 21 Jan, 2019, 06:37: AM
CBSE 11-science - Chemistry
Asked by Atulcaald | 25 May, 2018, 12:24: AM