ICSE Class 10 Answered
How do the infrared and 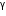 -rays differ in their following properties :
(i) wavelength, and (ii) penetrating power?
-rays differ in their following properties :
(i) wavelength, and (ii) penetrating power?
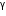 -rays differ in their following properties :
(i) wavelength, and (ii) penetrating power?
-rays differ in their following properties :
(i) wavelength, and (ii) penetrating power?
Asked by Topperlearning User | 21 May, 2014, 04:16: PM
(i) Although both the infrared and 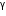 - rays are electromagnetic radiations,
- rays are electromagnetic radiations, 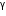 -rays have much shorter wavelength (
-rays have much shorter wavelength ( 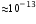 ) as compared to the infrared whose wavelength is nearly 10-6 m or more.
) as compared to the infrared whose wavelength is nearly 10-6 m or more.
(ii) 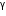 -rays are much more penetrating as compared to the infrared radiations.
-rays are much more penetrating as compared to the infrared radiations.
Answered by | 21 May, 2014, 06:16: PM
ICSE 10 - Physics
Asked by ayush421301 | 29 May, 2019, 11:08: PM
ICSE 10 - Physics
Asked by chumki.banerjee001 | 03 Mar, 2019, 07:10: PM
ICSE 10 - Physics
Asked by pcremyapc33 | 03 Mar, 2019, 12:06: PM
ICSE 10 - Physics
Asked by prodyumna02 | 04 Feb, 2019, 08:28: PM

