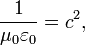CBSE Class 12-science Answered
where Q1 represents the quantity of charge on object 1 (in Coulombs), Q2 represents the quantity of charge on object 2 (in Coulombs), and d represents the distance of separation between the two objects (in meters).
Note: The symbol k is a proportionality constant known as the Coulomb's law constant. The value of this constant is dependent upon the medium that the charged objects are immersed in. In the case of air, the value is approximately 9.0 x 109 N • m2 / C2.
Imp: If the charged objects are present in water, the value of k can be reduced by as much as a factor of 80. It is worthwhile to point out that the units on k are such that when substituted into the equation the units on charge (Coulombs) and the units on distance (meters) will be canceled, leaving a Newton as the unit of force.
The proportionality constant k, called the Coulomb constant (sometimes called the Coulomb force constant), is related to defined properties of space and can be calculated based on knowledge of empirical measurements of the speed of light:In SI units, the speed of light in vacuum (or electromagnetic waves, in general), denoted c, which is equal to be 299,792,458 m·s−1, and the magnetic constant (μ0) is set at 4π × 10−7 H·m−1.
In agreement with electromagnetic theory, requiring that
the value for the electric constant (ε0) is derived to be ε0 = 1/(μ0c2) ≈ 8.85418782×10−12 F·m−1. In cgs units, the unit charge, esu or statcoulomb, is defined in such a way that the Coulomb constant is 1 and dimensionless.
We hope that clarifies your query.
Regards
Team
TopperLearning