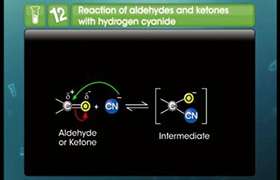
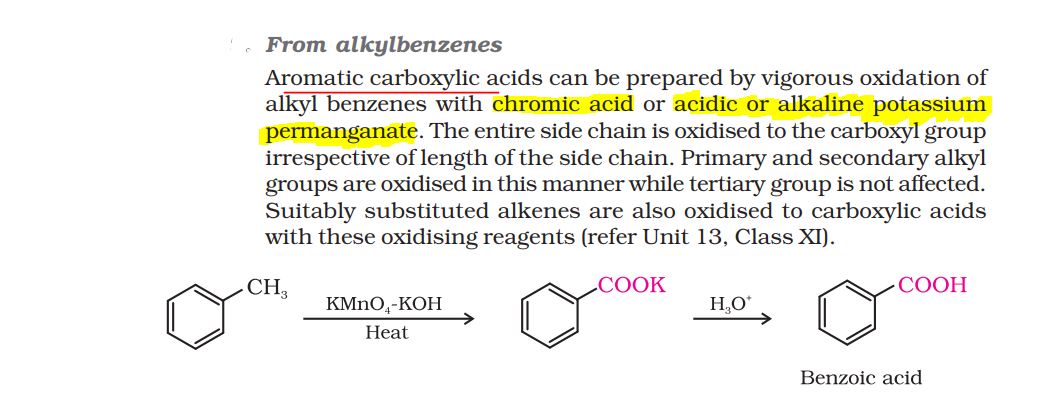
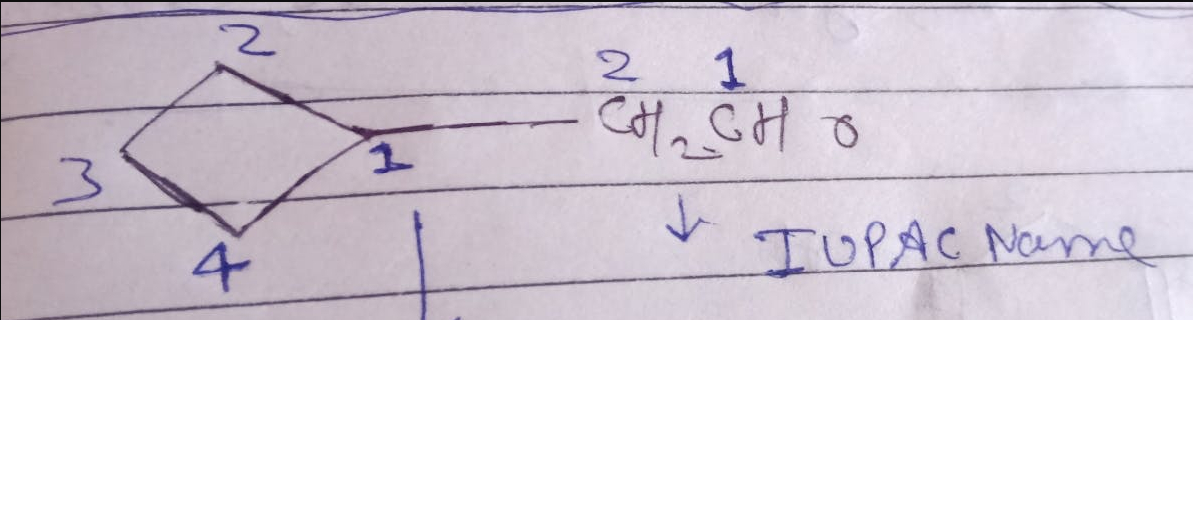
CBSE Class 12-science Answered
An oxidizing agent, or oxidant, gains electrons and is reduced in a chemical reaction. Also known as the electron acceptor, the oxidizing agent is normally in one of its higher possible oxidation states because it will gain electrons and be reduced.
A reducing agent, or reductant, loses electrons and is oxidized in a chemical reaction. A reducing agent typically is in one of its lower possible oxidation states and is known as the electron donor. A reducing agent is oxidized because it loses electrons in the redox reaction.
For example: Consider following reaction,
Cl2(aq) + 2Br− (aq) → 2Cl− (aq) + Br2 (aq)
Oxidation half reaction: 2Br− (aq) → Br2 (aq)
Reduction half reaction: Cl2 (aq) → 2Cl− (aq)
Br− loses an electron; it is being oxidized from Br− to Br2, thus Br− is the reducing agent.
Cl2 gains one electron; it is being reduced from Cl2 to 2 Cl−, thus Cl2 is the oxidizing agent.