CBSE Class 11-science Answered
Hello sir, please clarify this question
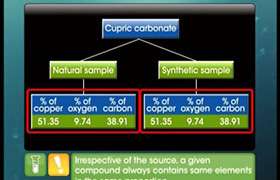
Does the law of multiple proportions hod true for Isotopes also? If not, why?
Please explain briefly.
Asked by seeni2005 | 08 Feb, 2021, 10:52: AM
Law of multiple proportions does not hold true for isoptopes. Because law multiple proportions states that when two elements combine to form two or more chemical compounds, then the masses of one of the elements which combine with a fixed mass of others, bear a simple ratio in one another.
Here, in case of isotopes, Atmoic masses are different so, masses are not fixed and ratio is going to be different.
Answered by Ravi | 08 Feb, 2021, 12:26: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by dhrubapratimc | 13 Sep, 2023, 09:34: PM
CBSE 11-science - Chemistry
Asked by abhiabhishek842006 | 09 Jan, 2023, 08:34: PM
CBSE 11-science - Chemistry
Asked by Shashisinghkusum | 13 Jul, 2022, 02:44: PM
CBSE 11-science - Chemistry
Asked by shivanshiarora3457 | 09 Jun, 2022, 03:28: PM
CBSE 11-science - Chemistry
Asked by rishamariyam222 | 28 Sep, 2021, 09:18: PM
CBSE 11-science - Chemistry
Asked by seeni2005 | 08 Feb, 2021, 10:52: AM
CBSE 11-science - Chemistry
Asked by manteaditya8 | 08 Jan, 2021, 01:45: PM
CBSE 11-science - Chemistry
Asked by muanputhomte4 | 11 Nov, 2020, 09:01: PM
CBSE 11-science - Chemistry
Asked by pradeepprince858 | 07 Aug, 2020, 12:54: PM
CBSE 11-science - Chemistry
Asked by seeni2005 | 28 Jul, 2020, 11:03: AM