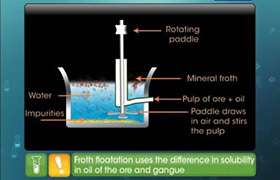
CBSE Class 12-science Answered
extraction of crude metal from concentrated ore
Asked by Febna Rasheed | 11 Mar, 2012, 09:49: PM
For a lot of metals (lead, mercury are good examples), the metal forms a weaker ionic interaction with a "hard" ion like oxide compared to a "softer" ion like sulfide. This weakens the ionic bonding in the solid and makes the metal ion more reactive.
Hence we convert the sulphides to oxides so that the extraction of the metals becomes easy .
Answered by | 12 Mar, 2012, 12:19: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by yashrajgharte24.12dgatl | 14 Oct, 2021, 10:13: PM
CBSE 12-science - Chemistry
Asked by utsavs858 | 05 Mar, 2021, 12:36: AM
CBSE 12-science - Chemistry
Asked by akhilspillai2005 | 08 Nov, 2020, 03:17: PM
CBSE 12-science - Chemistry
Asked by bindersaran3 | 04 Jun, 2020, 09:18: AM
CBSE 12-science - Chemistry
Asked by tribikramdash16 | 16 May, 2020, 06:51: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 28 Feb, 2020, 11:39: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 28 Feb, 2020, 11:39: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 28 Feb, 2020, 11:38: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 28 Feb, 2020, 11:38: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 28 Feb, 2020, 11:37: AM