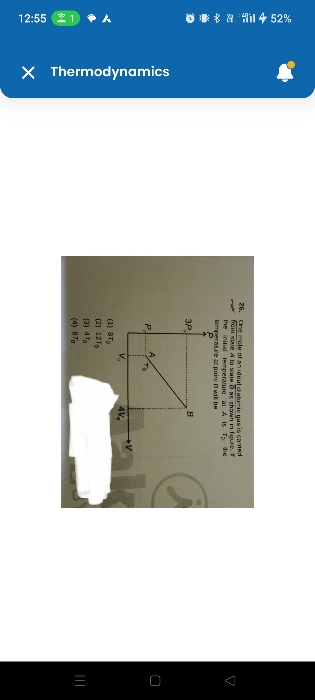
CBSE Class 11-science Answered
Internal energy, represented by E (or U a matter of preference), is essentially the thermal energy contained in a system (or particles making up the system). Unless change takes place, we usually have no way of evaluating it. A change in internal energy dE is due to the transfer of energy into or out of a system, but the volume stays constant. For example, energy transferred into the system, usually heat (q) and work (w), represents an increase of internal energy, dE, of the system. Thus,
In the case when heat or work is transferred from the system to its surroundings, the heat and work will be treated as negative quantities, resulting in a decrease in internal energy E.
The internal energy, E, does not depend on how energy is transferred and at what rate. It is purely an accounting of energy content of the system, and as such, the internal energy, E, is called a state function. The difference of a state function depends on the final and initial states, and we represent the change by,