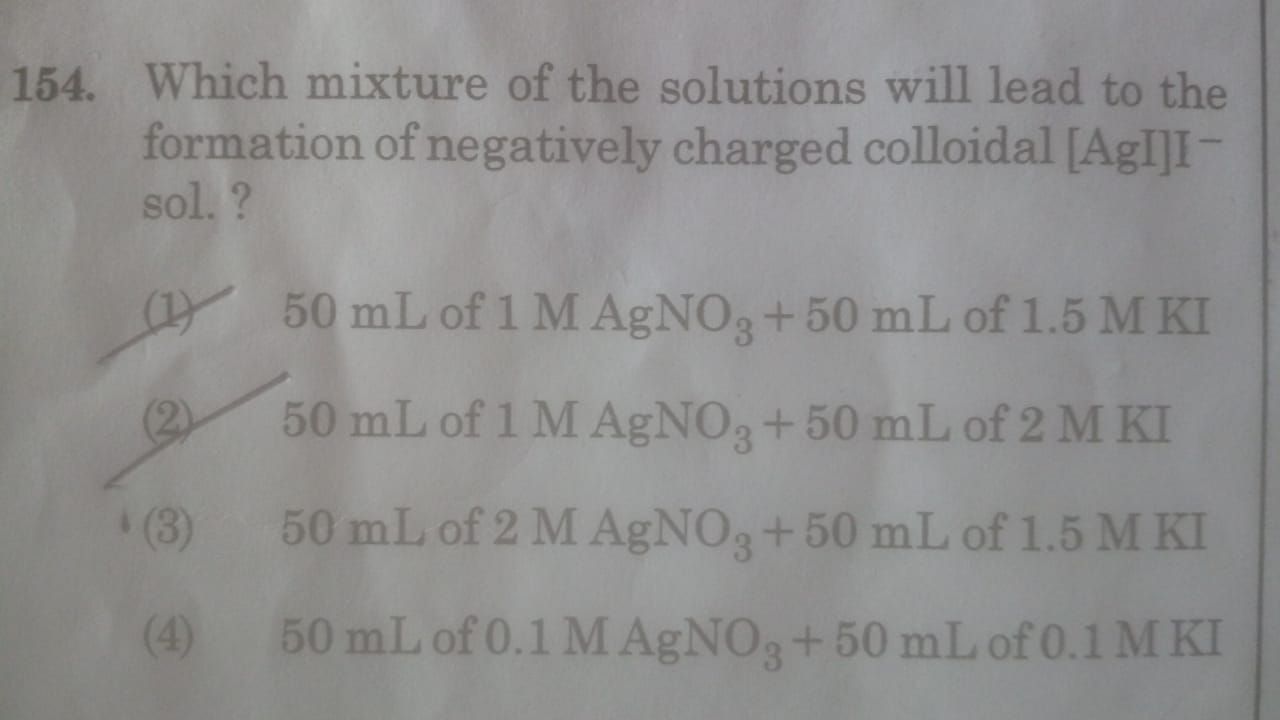CBSE Class 12-science Answered
catalysts speeds up the reaction, but it does not shift the position of the equilibrium. explain?
what equilibrium are they talking about?
Asked by Riya Satish | 17 Aug, 2010, 04:29: PM
For a given temperature, a catalyst will speed up the reaction and therefore speed up the attainment of the equilibrium, but will not alter the proportions of reactants and products
An equilibrium is able to shift in either direction, either towards reactants or towards products but since the proportion of reactants and products is not affected bt catalyst it does not shift the equilibrium
Answered by | 18 Aug, 2010, 10:43: AM
Application Videos
Concept Videos
CBSE 12-science - Chemistry
Asked by mallavaramchandrika6 | 29 Sep, 2021, 05:27: PM
CBSE 12-science - Chemistry
Asked by Akshij Nanda | 11 Mar, 2021, 10:21: AM
CBSE 12-science - Chemistry
Asked by sivakapuganti1 | 26 Aug, 2020, 08:57: PM
CBSE 12-science - Chemistry
Asked by spoorthysaienelluri | 23 May, 2020, 11:07: AM
CBSE 12-science - Chemistry
Asked by shreemuvijihari | 18 May, 2020, 06:29: PM
CBSE 12-science - Chemistry
Asked by vermahitesh124 | 12 May, 2020, 07:58: AM
CBSE 12-science - Chemistry
Asked by yogeshsulakh | 07 Feb, 2020, 09:28: AM
CBSE 12-science - Chemistry
Asked by tlb2bpartner | 21 Aug, 2019, 10:28: PM
CBSE 12-science - Chemistry
Asked by ntg432000 | 22 May, 2019, 08:10: PM
CBSE 12-science - Chemistry
Asked by kripanjalihimansu | 01 Mar, 2019, 04:16: PM