CBSE Class 12-science Answered
At a given instant there are 25% undecayed radioactive nuclei in a sample. After 10 sec, the number of undecayed nuclei is reduced to 12.5%.
calculate a) mean life of the nuclei b)the time in which the number of undecayed nuclei will further reduce to 6.25% of the reduced number.
Asked by Duwanie | 25 Nov, 2014, 12:20: AM

Answered by Romal Bhansali | 25 Nov, 2014, 10:43: AM
Concept Videos
CBSE 12-science - Physics
Asked by arjunsah797 | 16 May, 2022, 02:17: PM
CBSE 12-science - Physics
Asked by merinlijo_20002 | 17 Jun, 2020, 10:05: AM
CBSE 12-science - Physics
Asked by rajubarman | 01 Dec, 2019, 10:03: AM
CBSE 12-science - Physics
Asked by alanpeter9611 | 23 Feb, 2019, 07:47: PM
CBSE 12-science - Physics
Asked by alanpeter9611 | 22 Feb, 2019, 11:24: PM
CBSE 12-science - Physics
Asked by sd2021667 | 04 Dec, 2018, 04:46: PM
CBSE 12-science - Physics
Asked by silladech | 16 Nov, 2018, 11:14: AM
CBSE 12-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 09 Jul, 2014, 04:06: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 09 Jul, 2014, 04:15: PM






 , find the number of nuclei present after 8 days?
, find the number of nuclei present after 8 days?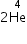 →
→  + ..............
+ ..............
