CBSE Class 12-science Answered
A solution containing 4 g of a non-volatile organic solute per 100 mL was found to have an osmotic pressure equal to 500 cm of mercury at 27oC27oC.The molecular weight of solute is:
A 14.97
B 149.7
C 1697
D 1.497
Asked by gangaram0 | 15 Jul, 2018, 02:21: PM
Option (B) is correct.
Given:
Weight of solute , W = 4 gm
Volume, V= 100 ml
V = 0.1 litre
Osmotic pressure, Π = 500 cm of Hg
Π = 6.57 atm
Tmpearture, T = 27 °C
T = 300 K
We know,
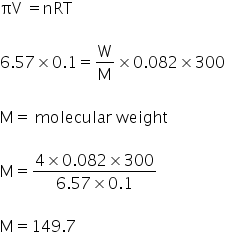
The molecular weight of solute is 149.7
Answered by Varsha | 15 Jul, 2018, 06:36: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by rashmij34 | 27 Feb, 2024, 04:42: PM
CBSE 12-science - Chemistry
Asked by premkhare2006 | 24 Jan, 2024, 09:50: AM
CBSE 12-science - Chemistry
Asked by kaushikmisty07 | 31 Dec, 2023, 11:42: AM
CBSE 12-science - Chemistry
Asked by KRISHPATEL.soc | 21 Jun, 2021, 05:58: PM
CBSE 12-science - Chemistry
Asked by dhrubajyoti.das | 09 May, 2021, 09:54: PM
CBSE 12-science - Chemistry
Asked by tiwariaatman | 31 Jul, 2020, 05:10: PM
CBSE 12-science - Chemistry
Asked by yogendrasoni142 | 08 Jun, 2020, 05:43: PM
CBSE 12-science - Chemistry
Asked by santosh357m | 28 Apr, 2020, 09:28: AM
CBSE 12-science - Chemistry
Asked by Balbir | 27 Jul, 2019, 05:02: PM
CBSE 12-science - Chemistry
Asked by ajaysankhala051 | 04 Jun, 2019, 02:28: PM






