CBSE Class 12-science Answered
A gas is obtained by the reaction of magnesium nitride with water. It acts as a lewis base and turns red litmus blue. It reacts with CuO as a reducing agent. It is obtained from two elements by reacting at 700K and high pressure. It is highly soluble in water. It forms blue coloured solution with copper sulphate.
Identify the gas and write all the reactions involved.
Asked by lekhakarthikeyan | 28 Aug, 2018, 03:52: AM
A gas is obtained by the reaction of magnesium nitride with water. It acts as a Lewis base and turns red litmus blue.
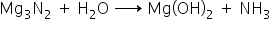
Magnesium nitrite reacts with water to produce gas ammonia, which turns red litmus blue.
Ammonia reacts with CuO as a reducing agent.
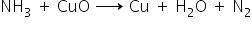
Ammonia is obtained from two elements, hydrogen and nitrogen by reacting at 700K and high pressure.
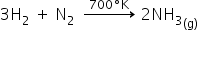
Ammonia forms a blue coloured solution with copper sulphate.
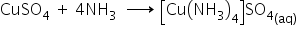
Answered by Varsha | 28 Aug, 2018, 03:31: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by shwetayaligar205 | 21 Dec, 2022, 07:45: PM
CBSE 12-science - Chemistry
Asked by kushwaharitik9129 | 14 Jul, 2022, 01:06: PM
CBSE 12-science - Chemistry
Asked by mufeedatvp2000 | 17 Apr, 2020, 09:22: AM
CBSE 12-science - Chemistry
Asked by adarshkamble130 | 19 Aug, 2019, 12:22: AM
CBSE 12-science - Chemistry
Asked by ranvirsingh1as | 16 Dec, 2018, 10:50: PM
CBSE 12-science - Chemistry
Asked by ranvirsingh1as | 16 Dec, 2018, 10:47: PM
CBSE 12-science - Chemistry
Asked by Sneha | 16 Dec, 2018, 03:14: PM
CBSE 12-science - Chemistry
Asked by lekhakarthikeyan | 28 Aug, 2018, 03:54: AM
CBSE 12-science - Chemistry
Asked by lekhakarthikeyan | 28 Aug, 2018, 03:52: AM
CBSE 12-science - Chemistry
Asked by niharikapabba2605 | 07 Aug, 2018, 01:00: AM





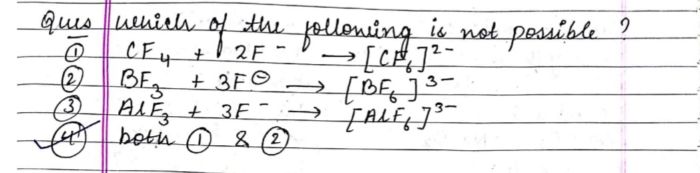
 to produce a brown coloured gass which intensifies on addition on copper turnings . on adding dilute ferrous sulphate olution to an aqueous solution to an aquaeous solution of X and then carefully adding conc.
to produce a brown coloured gass which intensifies on addition on copper turnings . on adding dilute ferrous sulphate olution to an aqueous solution to an aquaeous solution of X and then carefully adding conc. 